One most important electrical properties of the material is its ability to conduct electric current. This is decided by a set of properties which are collectively called Electrical properties of the material.
There are various materials that are used in the production of a certain item in an industry or a production unit. Therefore, the knowledge of the various physical and chemical properties of these materials is of utmost importance.
Fundamentally, there are two types of materials when classified according to their electrical conductivity – Conductors and Non-Conductors or Insulators. There is a third type known as Semi-conductors which have electrical conductivity lying between those of conductors and non-conductors. Whether a given material is a conductor or insulator can be judged based on the electrical properties of the material.
Electrical properties also give us an idea about the atomic buildup of a material. Metals, for example, have free electrons in their atomic structure. Electrons are charge carriers and hence metals are good conductors of electricity. So, we know that if something is a good conductor of electricity that means they have free electrons in the outer-shell of their atomic configuration. Similarly, we can draw various conclusions about a given material and compare them to find the best one for our own needs.
Let’s discuss some of the important electrical properties of a material.
Electrical Properties of Materials
The followings are the Electrical properties of engineering materials.
- Resistivity
- Conductivity
- Band Gap
- Dielectric Constant
- Magnetic Permeability
- Permeability
- Temperature Coefficient of Resistance
- Thermoelectricity
- Superconductivity
- Piezoelectricity
Now, we will discuss each property in detail.
1. Resistivity
Resistivity is one of the most important and fundamental electrical properties of a material. It can be defined as a measure of the resisting power of a given material to the flow of a certain amount of electric current. The higher the resistivity, the worse the material becomes in terms of electrical conductivity. Therefore, it goes without saying that good conductors have lower resistivity as compared to bad conductors or insulators.
Mathematically, resistivity is given by
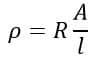
Where R is the resistance in Ohms, A is the area of cross-section in m^2 and l is the length in m of the material. Thus we can say that the resistivity of a material is the resistance offered to the flow of electric current by the material having unit length and unit area of cross-section.
The resistivity of a material is constant for a given temperature. It depends on the nature of the material and its temperature. Following is a table of the resistivity of different electrical materials at a given temperature.
| Material | Resistivity(Ohm-m) at 20 deg. C |
| Silver | 1.5 x 10-8 |
| Copper | 1.68 x 10-8 |
| Gold | 2.24 x 10-8 |
| Aluminum | 2.82 x 10-8 |
| Calcium | 3.36 x 10-8 |
| Tungsten | 5.60 x 10-8 |
| Zinc | 5.90 x 10-8 |
| Nickel | 6.99 x 10-8 |
| Iron | 1.00 x 10-7 |
| Lead | 2.20 x 10-7 |
| Nichrome | 1.10 x 10-6 |
| Carbon (Graphite) | 2.50 x 10-6 |
| Germanium | 4.60 x 10-1 |
| Drinking water | 2.00 x 10-1 |
| Silicon | 6.40 x 102 |
| Wet wood | 1.00 x 103 |
| Rubber | 1.00 x 1013 |
| Air | 1.30 x 1016 |
2. Electrical Properties of Materials- Conductivity
Conductivity is the electrical property that is reciprocal of resistivity which is a measure of ease of flow of electric current through a given material. It is measured in Siemens or Mho/meter (Mho=1/Ohm) and is given by
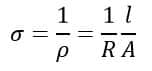
Good conductors of electricity have higher conductivity while it is low for bad conductors.
3. Band Gap
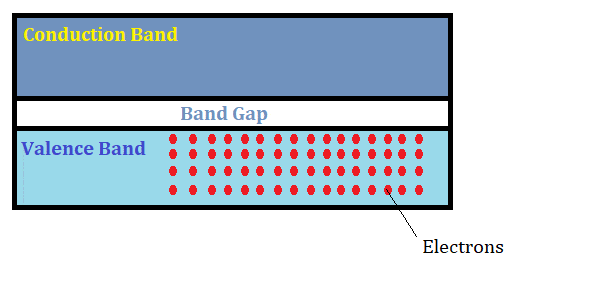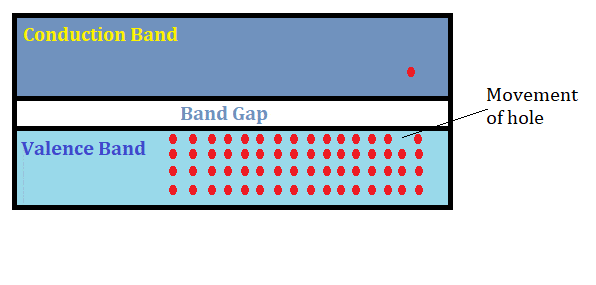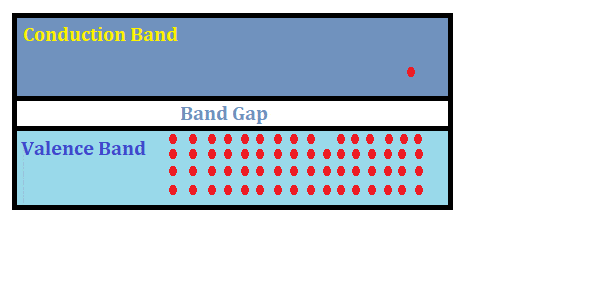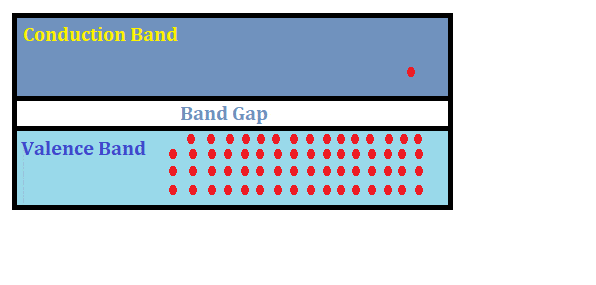
The band gap is an important electrical property of a material that tells us about the nature of the material at the atomic level. A band gap can be defined as the minimum amount of energy that is required to free an electron in the material so that it can participate in the process of conduction.
Any material has two energy bands – The Valence Band and The conduction band. The valence band is the low energy band where electrons are bounded by their respective nucleus. Band gap is the minimum amount of external energy that is required for the electron to jump to the nearest higher level of energy in the conduction band and therefore participate in the process of conduction.
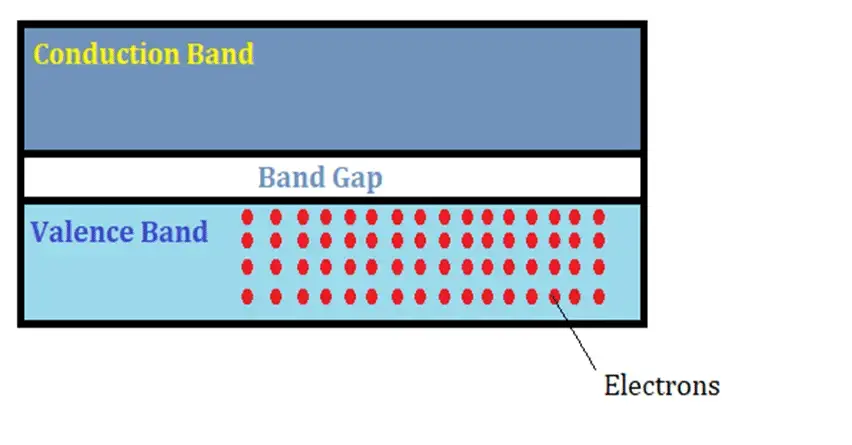
Based on the band gap energy property, electrical materials can be segregated as conductors, semiconductors, and insulators. Conductors have the lowest band gap as compared to semiconductors followed by insulators.
4. Electrical Properties of Materials-Dielectric Constant
The dielectric constant is an electrical property of the material and it is also known as the relative permittivity of a material. It is defined as the ratio of the permittivity of the substance to that of free space. It is a measure of the ability of a material to hold the electric flux within itself.
Mathematically, the dielectric constant is given by
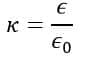
Where ε is the permittivity of the given material and ε0 is the permittivity of free space.
The dielectric constant is a ratio and hence it has no unit or dimension of itself. Its measurement is important for a material to be used for creating capacitance in a circuit or for the formation of an insulating material for a certain voltage or a range of voltage.
Following is a table of the Dielectric constant of various materials.
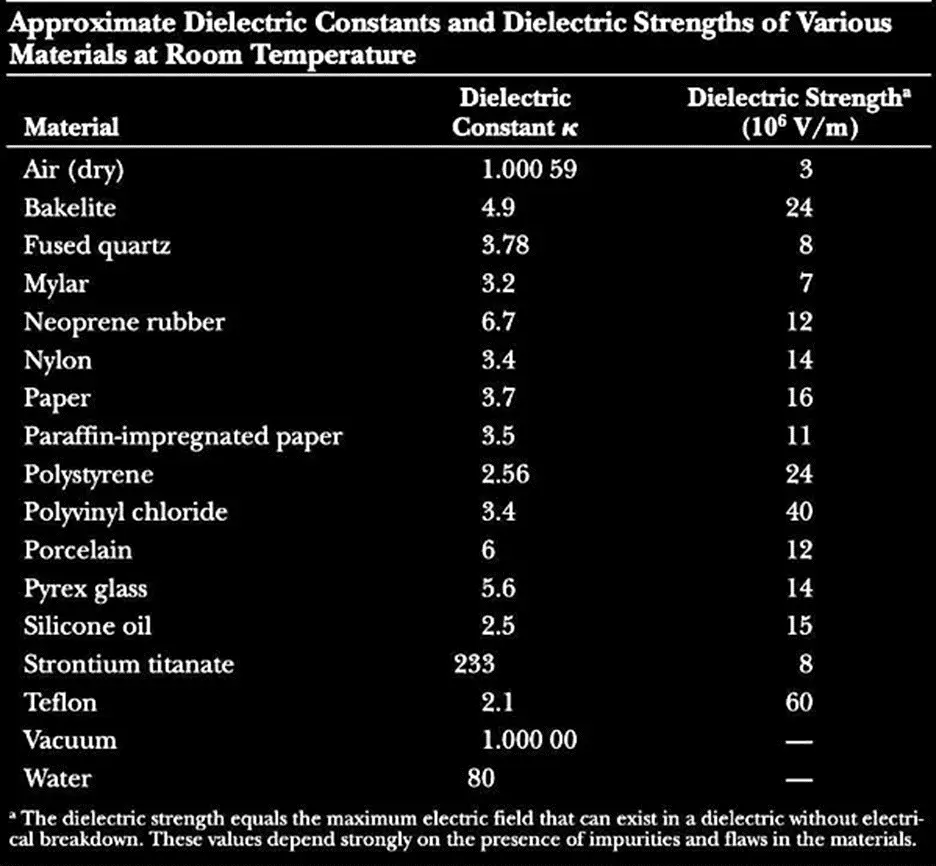
5. Magnetic Permeability
Magnetic permeability is an important electrical property of a material that quantifies or defines how the material in question responds to a magnetic field. It is the measure of the ease with which a material can be magnetized by a magnetic field in its vicinity.
Based on the magnetic permeability property, electrical materials can be classified as Ferromagnetic, paramagnetic and diamagnetic materials.
Ferromagnetic materials are the ones that are easily attracted by magnets. These materials are good conductors of magnetic field flux as the leakage flux through these materials is very small. Iron, Nickel, cobalt, CRGO steel, etc. are some examples. Ferromagnetic materials are used in the making of the magnetic core of a machine like a transformer or motor.
Paramagnetic materials are attracted to magnets but their affinity to magnets is feeble as compared to those of Ferromagnetic materials. These materials have impaired electrons. Viz: Lithium, Magnesium, Molybdenum, etc.
Diamagnetic materials are the ones that repel both poles of a magnet. Examples: Zinc, Silver, glass, Sodium Chloride, Antimony, etc.
6. Breakdown Voltage
The breakdown voltage of a material is the potential difference at which the material starts conducting electric current. This is not of much importance in the case of conducting materials but it is an important property to look at in the case of insulating materials. Because insulating materials can resist the flow of electric current across them up to a certain range of voltage only. This range can be determined by knowing the breakdown voltage of the material. The higher the breakdown voltage, the better the material becomes in terms of insulating ability.
7. Temperature Coefficient of Resistance
The electrical resistance of a material depends on the temperature of its surrounding. The temperature coefficient of resistance of a material is a constant value that indicates the rate of change of resistance with respect to temperature. It is denoted by α.
For a material let’s consider the resistance is R1 at a temperature of t1 deg. C and R2 at t2 deg.C. If the temperature coefficient of resistance of the material is α then,
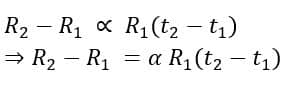
The unit of the temperature coefficient of resistance is per deg.C. The table below shows the value of the temperature coefficient of resistance of certain materials at 20 deg C.
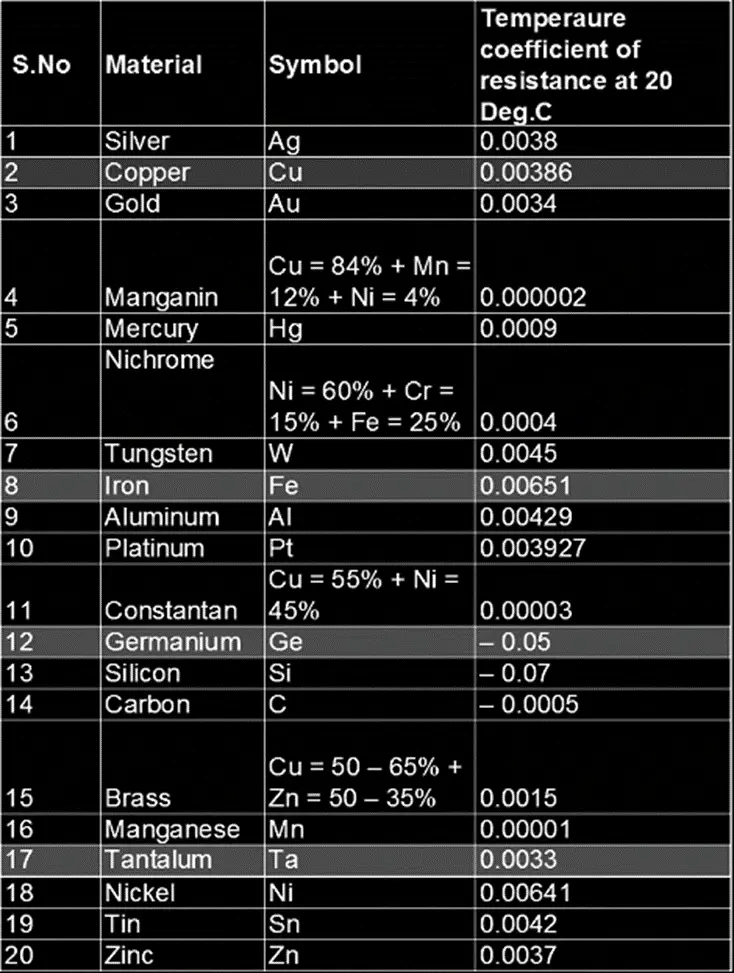
These are some of the important electrical properties of a material based on which their usage for a certain application is determined.
8. Thermoelectricity
When the junction formed by joining two metals is heated up, the voltage is induced at the open ends of the metals. This phenomenon is called the thermoelectric effect. The thermocouples, small power generators, and some transducers functions on the same principle.
9. Superconductivity
When material lies below the critical temperature, it offers zero electrical resistance. This property of the material is called superconductivity. The electrical conductivity of the materials becomes infinite when some materials are operated in a particular temperature zone.
10. Piezoelectricity
When the material experiences a strain or pressure, it outputs voltage. The property of the material that converts pressure or strain into electrical energy is called Piezoelectricity. Examples of piezoelectric materials are load cells, microphones, speakers, etc.
Read Next: