The main difference between a primary and secondary cell is that the primary cell is not rechargeable whereas a secondary cell is rechargeable.
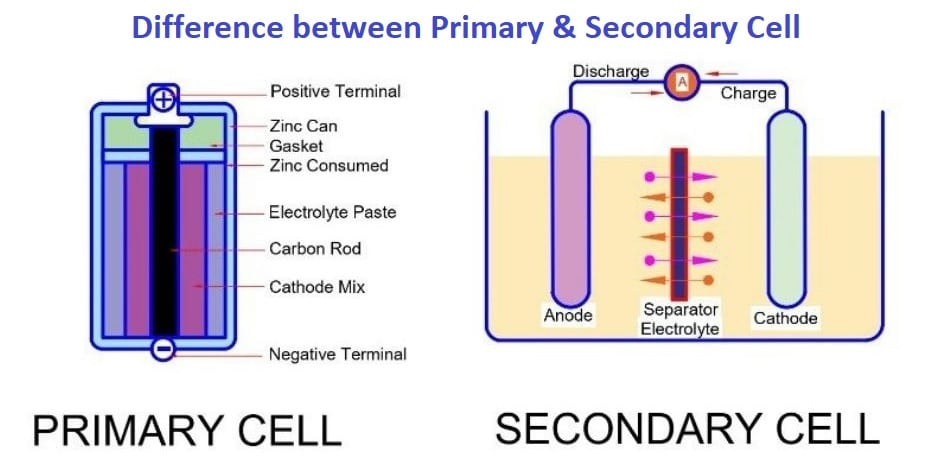
Primary cell
The primary cell has the following features.
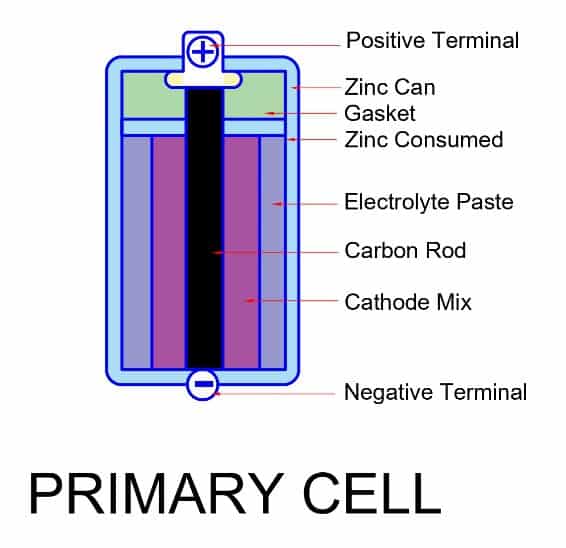
- It has high charge density. Therefore it discharges very slowly.
- It has high internal resistance. The high internal resistance cause more voltage drop during discharging. As a result, the terminal voltage is less than the EMF of the cell.
- There is no fluid inside the cell. That is why we also call it a dry cell.
- Its chemical reaction is irreversible. Therefore, it can not be charged again.
- It is cheaper than the secondary cell.
Secondary Cell
The secondary cell has the following features.
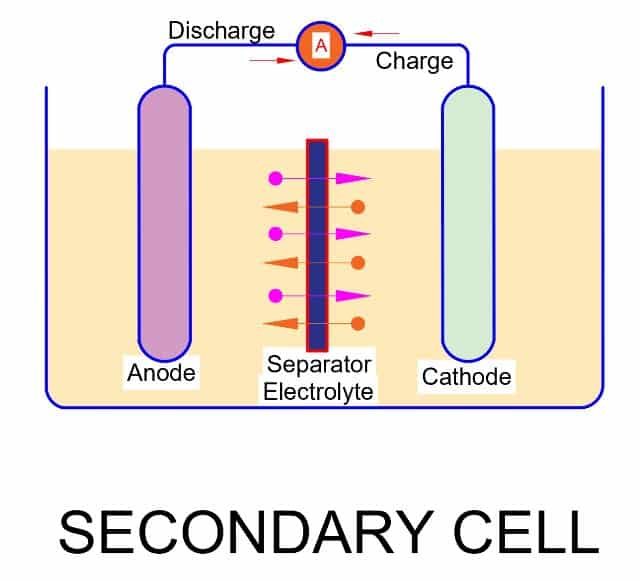
- It has low charge density. Therefore it discharges quickly.
- The secondary cell has low internal resistance. The low internal resistance cause less voltage drop during discharging of the cell. As a result, the terminal voltage is almost equal to the cell EMF.
- It has wet cells and molten salt. Therefore, the periodic maintenance is required.
- The chemical reaction in secondary cell is reversible. Therefore, it can be recharged. It is also called rechargeable battery.
- It is cheaper than the primary cell.
Difference Between Primary Cell and Secondary Cell
Both primary and secondary cells are useful for various applications. The primary cell does not reverse the chemical reaction after discharging. Therefore, it can not be reused. On the other hand, the secondary cell reverses the chemical reaction after discharging. Therefore, we can use it over & over again till its useful life exhaust.
The difference between the primary & secondary cells is as summarized in the below table.
| Primary Cell | Secondary Cell |
| The primary cell has an irreversible chemical reaction. Therefore, it can not be used again. | The secondary cell has a reversible chemical reaction. The same cell can be used again after recharging it. |
| Its internal resistance is high. Therefore, the terminal voltage is less. | Its internal resistance is low. Therefore, there is less voltage drop in the cell & terminal voltage is more while discharging. |
| It has a high energy density. | The cell has a low energy density. It can deliver the power for a lesser period of time & there is a need to recharge it. |
| The cell discharges slowly. | It discharges quickly. |
| No fluid inside the cell | It has wet cells and molten salt |
| Its design is simple. | The cell design is intricate. |
| Initial cost high | Initial cost low |
| Used only once | Can be used more than once |
| Examples of these cells are dry cells, daniel cells | Examples of these cells are lead-acid cells, Nickle-iron cells. |
Read Next