What is an Electrical Conductor?
An electrical conductor is a material that allows electric current to flow easily. Electric current is the flow of electric charges over time, and for this flow to happen, a medium is required—this medium is called a conductor.
The ability of a material to conduct electricity depends on the presence of free electrons. The more free electrons a material has, the better it conducts electricity, making it an effective conductor. Common examples include copper, aluminum, and silver.
To understand why some materials conduct electricity better than others, we need to look at the concept of free electrons.
Concept of free electrons
Free electrons are electrons in a material that are loosely attached to the nucleus. When an external electromotive force (EMF) or a time-varying magnetic field is applied, these electrons can move freely and allow electric current to flow.
Every atom has a nucleus with electrons orbiting around it. These electrons are grouped into two energy bands based on their binding strength:
- Valence Band Electrons – These electrons are tightly bound to the nucleus and do not participate in conduction under normal conditions. It requires a significant amount of energy to free them.
- Conduction Band Electrons – These electrons are loosely bound and can readily move under an applied EMF or magnetic field, making them responsible for electrical conduction.
In electrical conductors, the conduction band and the valence band almost overlap, which allows electrons to move easily from one band to another. This is why conductors like copper and aluminum have high electrical conductivity.
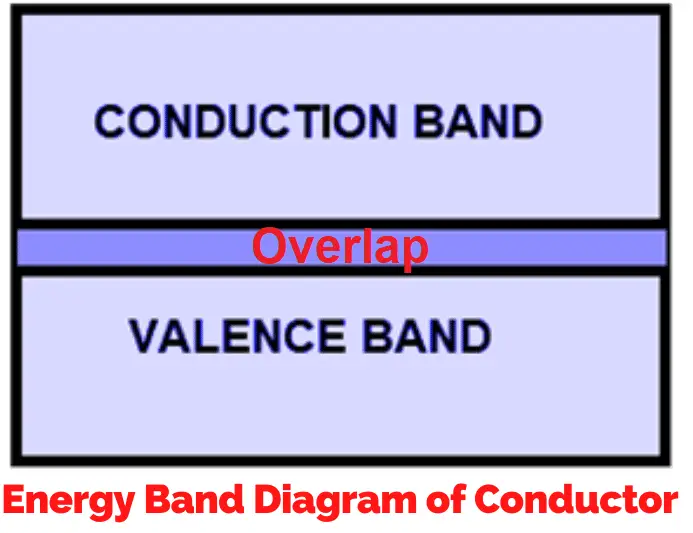
At the atomic level, the movement of electrons between the valence and conduction bands explains how materials conduct electricity. In theory, if a material allowed electrons to move without any resistance, it would represent the perfect conductor. This brings us to the concept of an ideal conductor.
What is an Ideal Conductor?
An ideal conductor is a material with zero resistance and infinite conductance, allowing electric current to flow without any voltage drop. At the atomic level, electrons in an ideal conductor can move from the valence band to the conduction band without requiring any energy.
In the real world, however, every material has some inherent resistance, which depends on temperature and other physical conditions. When an electromotive force (EMF) is applied, current flows through the conductor, but a small voltage drop occurs due to this resistance.
Even voltage sources, like batteries, have internal resistance, which leads to a slight voltage drop across their terminals during current flow.
Properties of Electrical Conductors
Electrical conductors have several key properties that make them effective at carrying electric current:
- Free Movement of Charges – A conductor allows the movement of free electrons or ions within it, which is essential for current flow.
- Zero Internal Electric Field – The electric field inside a conductor is zero, enabling electrons to move freely without internal resistance.
- Charge Neutrality – Positive and negative charges inside a conductor are equal and cancel each other, resulting in zero net charge density within the material.
- Surface Charge Distribution – Free charges only exist on the surface of a conductor, not inside the bulk material.
- Perpendicular Electric Field – Any electric field at the surface of a conductor always acts perpendicular to the surface.
Read detailed article on: Properties of Electrical Conductor
Types of Conducting Materials
Electrical conductors are categorized based on their ability to conduct electricity. The three main types are good conductors, insulators, and semiconductors. Let’s look at each in detail.
Good Conductors
Good conductors are materials that allow electric current to flow easily due to the abundance of free electrons. These materials are widely used in electrical systems for the conduction of electricity.
Most metallic elements are good conductors, including iron, copper, silver, and nickel. They require very little energy to move electrons from the valence band to the conduction band.
While silver is the best conductor of electricity, it is expensive and not practical for most electrical applications. Therefore, copper is commonly used in electrical machines because it offers a good balance of conductivity, ductility, and malleability. Aluminum is preferred in applications where weight is a concern, such as in transmission lines.
Insulators (Bad Conductors)
Insulators, also known as bad conductors, are materials that have very few or no free electrons. They require a large amount of energy to move electrons from the valence band to the conduction band, often supplied by a high potential difference ranging from a few kilovolts to several megavolts. Because of this, insulators are not used for conducting electricity.
These materials are primarily used as electrical insulation to ensure safe handling of conductors and to provide isolation between circuit elements or between a circuit and its surroundings. This prevents accidents and ensures safe operation of electrical systems.
- Examples: Plastic, PVC, Bakelite
- Usage: Bakelite is commonly used in electrical switches due to its insulating properties.
In terms of bonding, electrical conductors form electrovalent bonds, while insulators typically form covalent bonds.
Semiconductors
Semiconductors are materials that are partially conductive, meaning they can behave like a conductor or an insulator depending on conditions such as temperature, applied voltage (EMF), or doping.
- Examples: Silicon, Germanium, Gallium Arsenide
- Key Use: Semiconductors are widely used in electronic circuits because they are ideal for fast-switching applications. Many electronic components, such as transistors and diodes, are made from semiconducting materials.
How does a Conductor Conduct Current?
As discussed earlier, conductors have a large number of free electrons that allow electric current to flow. When a potential difference is applied across two points in a conductor, these electrons gain energy and move from the low-potential region to the high-potential region in the conduction band. This movement of electrons from one atom to another is what constitutes electric current.
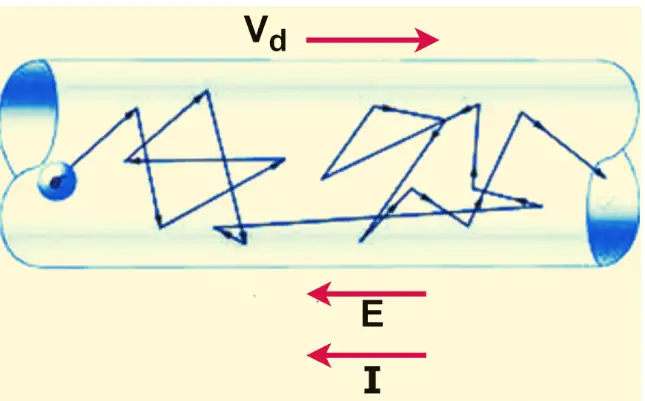
Since a conductor contains an enormous number of electrons (often called an electron cloud), the electrons do not move in a straight path. Instead, they perform a to-and-fro motion, frequently colliding with other electrons and atoms. The average velocity with which the electrons drift in the direction of the electric field is called the drift velocity. For a given material and electric field intensity, the drift velocity remains constant.
let’s look at drift velocity and how electrons actually move in a conductor
Drift Velocity
Drift velocity is the average velocity at which an electron moves forward under the influence of an electric field. Although millions of electrons are moving in a conductor, each electron frequently collides with other electrons and atoms. As a result, the velocity of an individual electron constantly changes. By taking the average of these random movements, we get the drift velocity of the electrons.
Therefore, drift velocity is given as
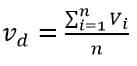
Where n is the number of collisions. Now we know Newtonian mechanics
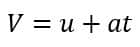
Here, ‘u’ represents the initial velocity of the electron, and ‘a’ is the acceleration. Between two successive collisions, the electron is influenced only by the electric field (E). Its initial velocity at this moment is the velocity it had just after the previous collision, and ‘t’ is the time elapsed since that collision.
Therefore,
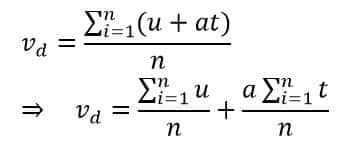
The first term in the drift velocity equation represents the average of all the initial velocities of the electrons. Because there are millions of electrons constantly colliding with each other, the vector sum of all their initial velocities averages out to zero.
The acceleration ‘a’ is a uniform acceleration given by
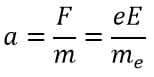
Here, F is the force acting on the electron, which is equal to the electric field force calculated as the product of the electron’s charge (e) and the electric field intensity (E). mₑ represents the mass of the electron.
Therefore,
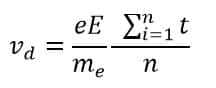
Now,
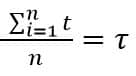
Here, τ (tau) is the relaxation time of the electron, which is the time interval between two successive collisions. Using this, the drift velocity of electrons in a conductor can be expressed as:
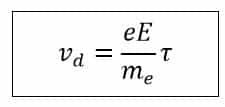
Thus, the drift velocity is a constant value for a given material, determined by the electric field intensity and the electron’s relaxation time (τ).
With the drift velocity understood, we can now examine the direction of electric current flow in a conductor.
Read detailed article on: Drift Velocity: Definition, Formula, Derivation & Solved Examples
Direction of flow of electric current in a conductor
When a conductor is subjected to a potential difference, an electric field is established within it. Electrons flow opposite to the direction of the electric field, while the electric current is defined as the flow of positive charge and moves from higher potential to lower potential, opposite to the electron flow.
The rate at which charge flows through a conductor over time is called electric current. Hence, the electric current can be expressed as:
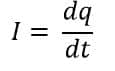
Now that we understand how electric current flows in a conductor, let’s look at the different types of conductors based on how they behave under applied voltage.
Read detailed article: Direction of Electric Current: Conventional Current vs Electron Flow
Types of Conductors
Ohmic Conductors
Ohmic conductors are materials that follow Ohm’s law, meaning the voltage (V) across them is directly proportional to the current (I) flowing through them.
- Examples: Iron, Copper, Silver, etc.
- Key Feature: Ohmic conductors do not produce harmonics in the electrical power system, ensuring a stable and clean supply of electricity.
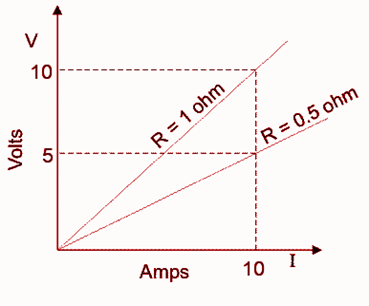
Non-Ohmic Electrical conductors
Non-Ohmic conductors are materials that do not obey Ohm’s law, meaning their voltage and current relationship is not linear. This behavior is typically seen in semiconductors.
- Examples: Diodes, Transistors, LDRs (Light Dependent Resistors)
- Impact: Non-Ohmic conductors can generate harmonics in the electrical power system, which may distort and pollute the power supply.
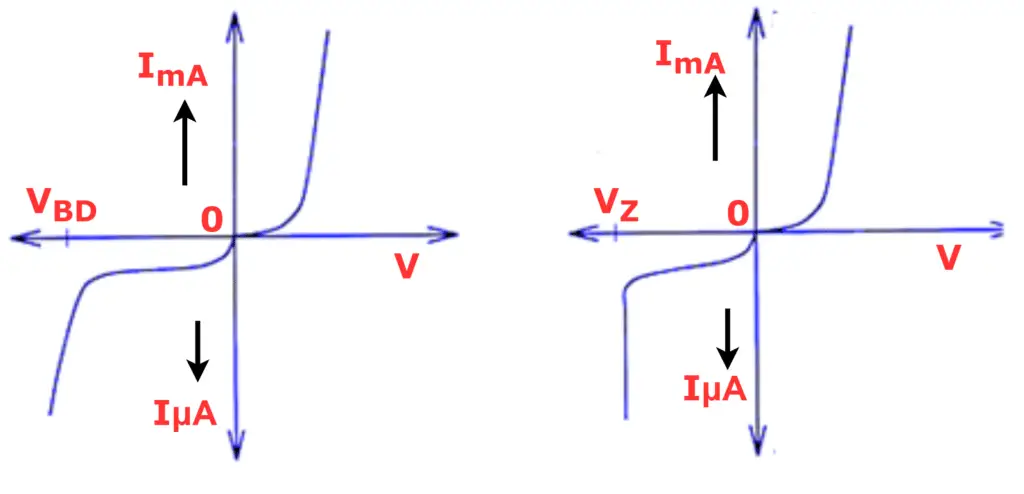
Read detailed article on: Difference Between Ohmic and Non-Ohmic Conductors
Examples of Electrical Conductors
Solid electrical Conductors
Solid conductors are materials in a solid state that allow the flow of electric current. Examples include:
- Metallic Conductors: Silver, Copper, Aluminum, Gold, etc.
- Non-Metallic Conductors: Graphite
- Alloy Conductors: Brass, Bronze, etc.
These materials are widely used in electrical systems due to their reliable conductivity and stability.
Liquid Electrical Conductor
Liquid conductors are materials in a liquid state that can conduct electricity. Examples include:
- Metallic Conductors: Mercury
- Non-Metallic Conductors: Saline water, acid solutions, etc.
These conductors are commonly used in electrolytic cells, batteries, and chemical processes where a liquid medium is required for current flow.
Effect of Temperature on a Conductor
As the temperature of a conductor increases, its molecules vibrate more intensely. These vibrations create obstacles for the moving electrons, reducing the overall conductivity of the material.
Although higher temperatures can sometimes release a few additional electrons by breaking molecular bonds, the increase is minimal. As a result, the electrical conductivity of the conductor still decreases with rising temperature.
Read detailed article on: Effect of Temperature on Conductors, Semiconductors & Insulators
Applications of Electrical Conductors
Electrical conductors play a vital role in transmitting and utilizing electric current across a wide range of applications in both everyday life and industrial systems. Some of the most notable uses include:
- Electrical Wiring:
Copper and aluminum conductors are commonly used in electrical wires to carry current from power sources to devices. They are insulated to prevent short circuits and ensure safety. - Electrical Circuits:
Conductors form the backbone of electrical circuits, connecting components like resistors, capacitors, inductors, diodes, and transistors to perform specific functions. - Electrical Machines:
Devices such as motors, generators, and transformers rely on conductors in their coils, windings, rotors, and stators to convert electrical energy into mechanical energy or vice versa. - Electrical Sensors:
Conductors are essential in sensors that detect physical quantities like temperature, light, pressure, or sound, converting them into measurable electrical signals. Examples include thermocouples, photodiodes, LDRs, and piezoelectric sensors. - Power Transmission and Distribution:
High-voltage transmission lines use aluminum or copper conductors to efficiently transport electricity over long distances from power plants to homes and industries.
Conclusion
Electrical conductors are indispensable materials for a wide variety of applications involving electric current. Their types, properties, and conductivity determine their suitability for specific uses—from simple wiring and circuits to advanced electrical machines and sensors. As technology advances, conductors continue to evolve, meeting the growing demands of modern electrical and electronic systems.

Related Articles:
- What is the difference between Drift Current and Diffusion Current?
- Difference Between Valence Band and Conduction Band
- Electric Current and Theory of Electricity
- Temperature Effect on Conductors, Semiconductors & Insulators
- Drift Velocity: Definition, Formula, Derivation & Solved Examples
- Relation Between Current and Drift Velocity – Formula, Diagram & Derivation
- Direction of Electric Current: Conventional Current vs Electron Flow
- Difference Between Ohmic and Non-Ohmic Conductors
- Electrical Stranded Conductor
- ACSR Conductor Types, Properties and its Advantages
- 10 Examples of Electrical Conductors and Insulators
- Good and Bad Conductors of Electricity Explained Simply