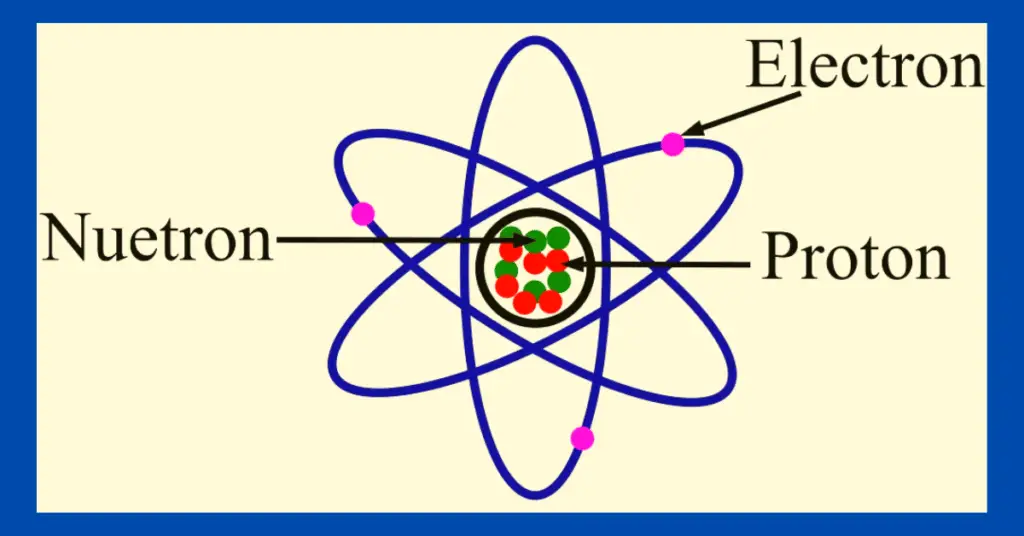
In this article, we will discuss the important differences between electron and proton. But before comparing electrons and protons, let us first know a bit about electrons and protons individually.
What is Electron?
Electron is an elementary subatomic particle that bears a negative electric charge. In other words, an electron is one of the fundamental particles of an atom that has a negative charge. Since an electron carries an electric charge, hence it is called a particle of electricity. In an atom, electrons are present in the space external to the nucleus (called extra-nucleus). In the extra-nucleus, electrons revolve in different paths called orbits. Each orbit has a fixed number of electrons which is called the electron configuration of the orbit.
In the graphical representation, the electron is generally represented by the small case letter “e”. It is a negative charge particle, whose charge value is,

The mass of an electron is,

Due to this small mass, the electron can easily move from one atom to another when some external energy is supplied to it. Based on the energy level, electrons can be classified into two types namely, valance electrons and conduction (free) electrons.
Valance electrons are those that are present in the outermost shell or orbit of the atom. While the conduction electrons are those that participate in current conduction, these electrons are free to move from one atom to another. Hence, conduction electrons are also known as valance electrons.
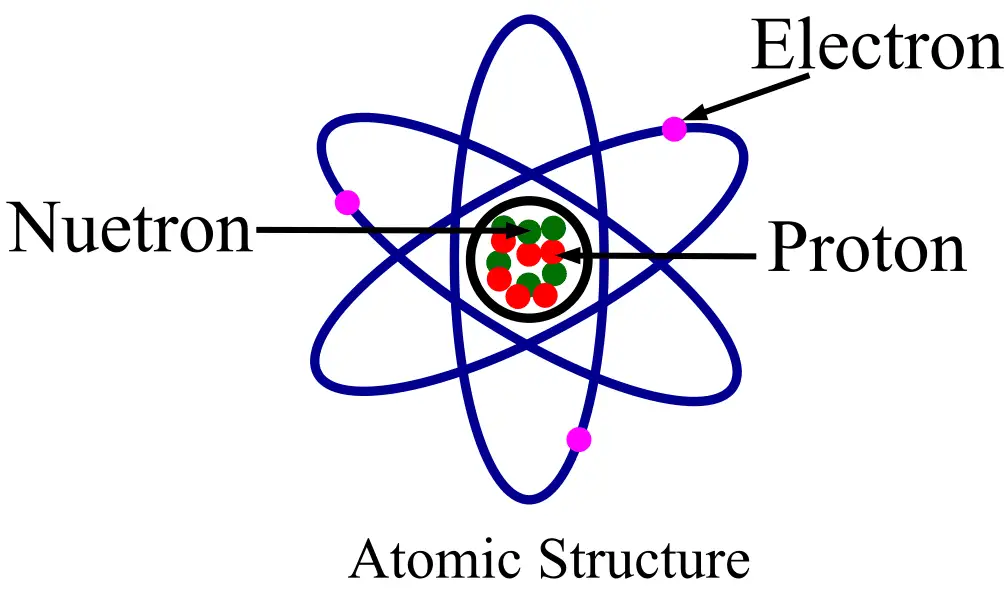
What is Proton?
Proton is also a type of subatomic particle. Proton is a positive charge particle that is present inside the nucleus of the atom. Therefore, a proton is attached firmly to its atom by nuclear forces and cannot leave the atom easily.
Similar to an electron, a proton also carries an electric charge, hence it is also called a particle of electricity, but it cannot participate in current conduction due to its immovable nature.
On the atomic diagram representation, a proton is represented by the symbol “p”. The charge on the proton is equal to the charge on an electron, but of positive polarity, i.e.

The proton is a heavier particle compared to an electron, the mass of a proton is,

The total number of protons in an atom is known as the atomic number of the element. Being a positively charged particle, the proton produces an electric field from the atom.
After discussing the basics of electron and proton, let us now discuss the differences between them.
Differences between Electron and Proton
The major differences between electron and proton are given in below table-
| Key | Electron | Proton |
| Definition | Electron is a subatomic particle of an atom that carries a negative charge. | Proton is a subatomic particle of an atom that carries a positive charge. |
| Symbolic representation | Electron is represented by the symbol “e”. | Proton is represented by the symbol “p”. |
| Nature of the charge carried | Electron carries a negative electric charge. | Proton carries a positive electric charge. |
| Magnitude of charge | The magnitude of electric charge carried by an electron is, Qe=-1.6 X 10-19 C | The magnitude of electric charge carried by the proton is, Qp=+1.6 X 10-19 C |
| Mass | The mass of an electron is, me=9.1 X 10-31 Kg | The mass of a proton is, mp=1.67 X 10-27 Kg Hence, a proton is comparatively heavier than an electron. |
| Direction of electric field | The electric field is directed toward the electron. | The electric field is directed away from the proton. |
| Participation in current conduction | Electrons are entirely responsible for the conduction of current in metallic conductors. | Protons do not participate in current conduction in conductors. |
| Presence in atomic structure | Electrons are present in the extra-nucleus (i.e. outer space around the nucleus) of the atom. | Protons are present inside the nucleus of the atom. |
| Classification | Based on energy levels, electrons are classified into two categories namely, valance electrons and conduction electrons. | There is no such classification of protons. |
| Significance | Electrons are responsible for the conduction of current, making bonds, participation in reactions, etc. | Protons are responsible for constituting the weight of an atom, balancing the charge on the atom, deciding an atomic number of elements, etc. |
Conclusion
Hence, this is all about the key differences between electron and proton. Though, both electron and proton are the subatomic particles of an atom, and are particles of electricity. But they are completely different from each other. From the above comparison, we may state the most significant difference between electron and proton that is, the electron is a negatively charged particle which present external to the nucleus of an atom, while a proton is a positively charged particle that is present inside the nucleus of the atom.