In this article, we will discuss the conductivity measurement methods. The contacting conductivity & inductive conductivity methods are two methods for conductivity measurement.
What is Conductivity?
- Conductivity is the measure of the ability of any solution to pass an electrical current.
- Conductivity is usually affected by free ions which are dissolved in any solution.
- The ions can be chloride, sulfate, nitrate, phosphate, sodium, magnesium, iron, calcium, or aluminum.
Is conductivity affected by temperature?
- Yes, conductivity is affected by temperature.
- Conductivity is directly proportional to the temperature of the solution.
- An increase in temperature will increase conductivity, and a decrease in temperature will decrease conductivity.
- The reason for this direct proportionality is that as we increase temperature, the kinetic energy of the ions present in the given solution also increases, and they start moving fast. This fast movement of ions causes an increase in conductivity and the inverse is the case in decreasing temperature.
- Hence, temperature compensation is needed for every conductivity meter to get accurate readings.
- Generally, conductivity is calculated based on the reference temperature of 25 degrees centigrade.
How do we measure conductivity?
There are two types of conductivity measurements: contact and induction. The choice to use depends on the conductivity’s magnitude, the fluid’s corrosive nature, and the number of suspended solids. In general, the induction method is better when the conductivity is high, the liquid is corrosive, or suspended solids are present. The two methods of conductivity measurement are;
- Contact conductivity measurement
- Inductive Conductivity Measurement
We will now discuss the conductivity methods in detail.
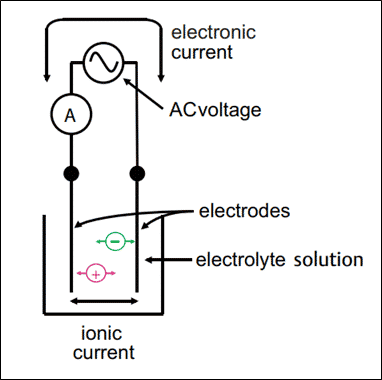
1. Contacting Conductivity Measurement
- The contacting conductivity probes consist of two electrodes made of metal (stainless steel or titanium) that are in contact with the given solution whose conductivity we need to measure.
- The contacting conductivity analyzer applies an alternating voltage to both electrodes.
- The electric field causes the ions in the solution to move back and forth, creating an electric current.
- Since the charge carriers in the solution are ions, the current is also called the ionic current.
- The analyzer uses Ohm’s law to calculate the resistance of the given solution (resistance = voltage/current).
- R = V / I
Where, R = Resistance
V = Voltage
I = Current
- Conductance is the inverse of Resistance
G = 1 / R
- The conductivity meter measures the conductance and then gives conductivity by converting the measured conductance.
- A term called cell constant comes into the picture to convert conductance into conductivity.
- The cell constant is the ratio of the distance between the plates of electrodes and the area covered by the electrodes.
- K = d / a
- K = cell constant
- d = distance between electrodes
- a = area covered by the electrodes
- Now conductivity is the product of conductance, and the cell constant
- Conductivity = G * K

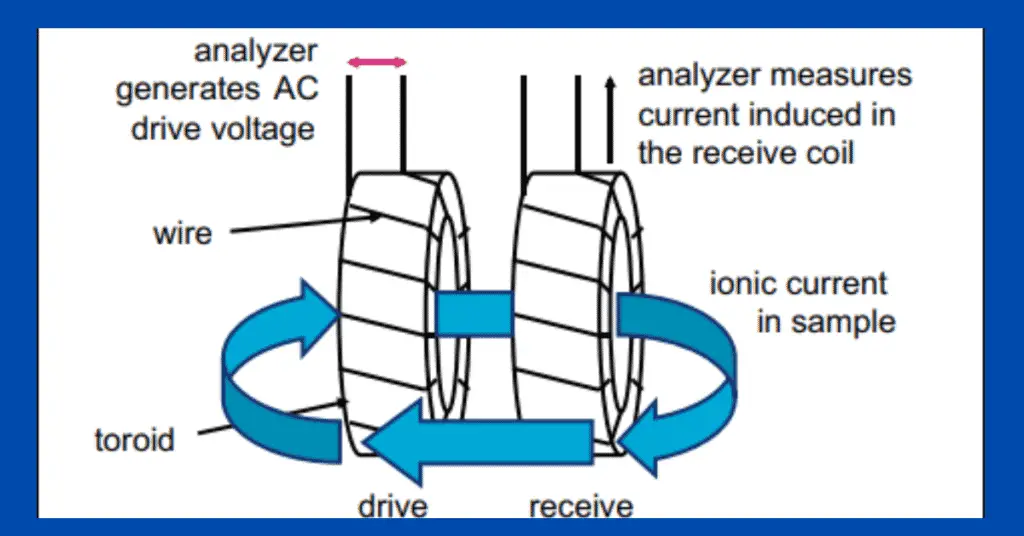
2. Inductive Conductivity Measurement
- Inductive conductivity meters are also called toroidal conductivity meters or electrodeless conductivity meters.
- Inductive conductivity meters consist of 2 metal coils inside a corrosion-resistant casing (usually plastic).
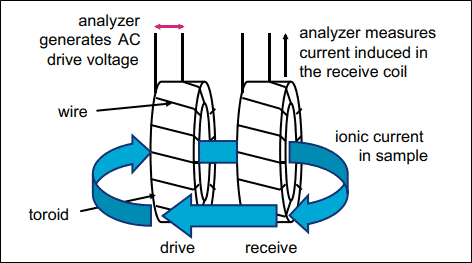
- Out of 2 coils, one is the drive coil, which generates alternating voltage, and the other is the receiving coil.
- The construction is like that of a transformer. The only difference is that the solution whose conductivity is to be measured behaves like a core.
- The voltage produced from the drive coil induces a voltage in the receiving coil, and the current starts flowing in proportion to the conductivity of the solution.
- This induction of voltage and current in the receiving coil is directly proportional to the conductivity of the solution.
- Inductive conductivity meters work only with solutions whose conductivity is more than 15 micro Siemens/centimeters.

Applications of conductivity measurement
- RO treatment water
- For desalination plant
- In boiler feedwater
- Cooling tower Water
- Hardness measurement in laundries.
- Condensate & steam quality protection
- Wastewater treatment
Factors affecting conductivity
- The concentration of the solution
- Mobility of ions in the solution
- The valence of ions in the solution
- The temperature of the solution
- Resistance of the cable
- Cell constant (cell constant keeps on changing due to the aging effect on the electrodes and also deposition on the electrodes)
How to Improve the performance and lifetime of the Conductivity meter?
- To maximize the performance and lifetime of your conductivity meter, it is important to clean and validate/calibrate the conductivity probe regularly because depositions can affect it badly.
- If your conductivity probe is not in use, then the probe should be stored in the recommended demineralized water storage solution or kept dry as per manufacturer recommendations.
- If your conductivity probe exhibits slow settling times, erratic readings, or difficulty in calibration, it may be time to clean the conductivity probe.
- Always clean the conductivity probe with demineralized water or other solution recommended by the manufacturer.
- Periodically calibrate the conductivity meter.
- One very important factor that leads to inaccurate readings is the temperature compensation in conductivity meters. Generally, all conductivity meters have an inbuilt temperature sensor (RTD) inside the conductivity probe for temperature compensation because conductivity is a temperature-dependent parameter. Therefore, temperature monitoring is a must for accurate measurement readings.
- Always verify the cell constant provided with the conductivity probe (usually written on the probe as shown in the picture below) and the one configured in the conductivity transmitter before using it.