The conductivity cell constant of the conductivity sensor is a multiplier constant. The cell constant is multiplied by the measured current to calculate the electrical conductivity of the solution.
A conductivity analyzer is mainly used in water treatment or wastewater treatment plants. The conductivity analyzer is one of the most fundamental analyzers for determining the quality of the water after certain processes like desalination plants, demineralized plants, and many more. The conductivity analyzer is even used to measure the conductivity of different liquids used in industrial applications to know their quality and contents.
A conductivity analyzer measures the conductivity of any given liquid. The sensor of the conductivity analyzer is immersed in the liquid whose conductivity is to be measured. The conductivity analyzer consists of two main parts. One is the transmitter of the conductivity analyzer, and the other is the sensor of the conductivity analyzer.
Today, in this article, we will have a detailed look into the types of contact conductivity analyzers. Now, one might think there is only one type of conductivity sensor. But no, there are different types of conductivity sensors. These sensors are differentiated on the basis of their cell constants.
What is the Conductivity Cell Constant?
We have seen different conductivity analyzers with different cell constants. So, what is this cell constant? The cell constant is nothing but a value dependent on the conductivity sensor’s area or size. The length between the sensor’s electrodes and the area of the electrodes determine the cell constant.
The cell constant of the conductivity sensor is directly proportional to the distance between the electrodes and inversely proportional to the surface area of the conductivity sensor’s electrodes. To get the conductivity sensor’s cell constant, divide the value of the distance between the electrodes by the surface area of the conductivity sensor’s electrodes. Hence, the unit of the cell constant is also cm-1 or per centimeter.
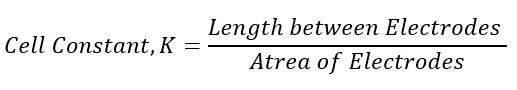
Hence, if one conductivity sensor has a distance between the plates of 1 cm and the area covered by the electrodes is 10 cm2, then to get the cell constant, we use:
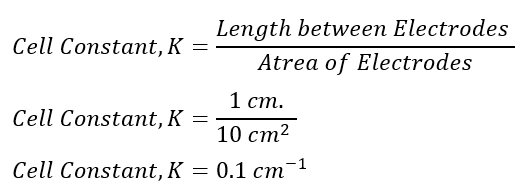
The cell constant is 0.1 cm-1
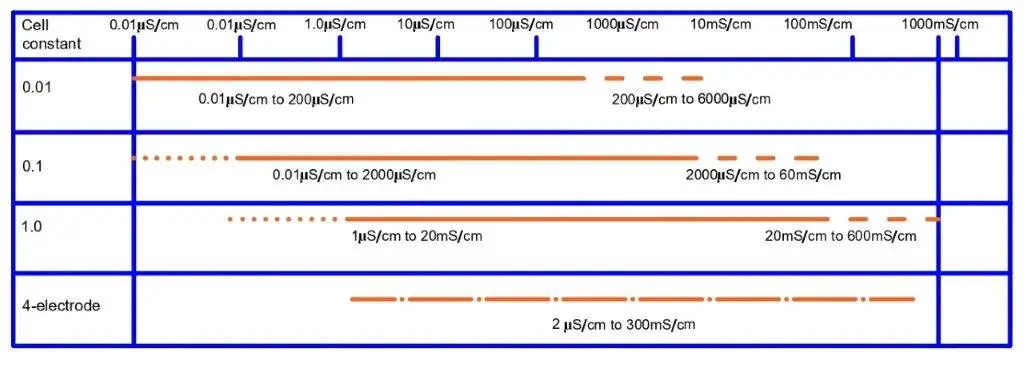
Similarly, we have seen conductivity sensors with different cell constants like 0.01, 0.1, 1, 10, and many more.
Why do we need Constants?
The reason behind this is that all liquids have different conductivities. For example, seawater can have a conductivity near 50000 µS/cm; raw water can have a conductivity near 10000 µS/cm; freshwater can have a conductivity near 1000 µS/cm; tap water can have a conductivity near 500 µS/cm, and distilled or pure water can have conductivity below 1 µS/cm. This is why a wide range of conductivity sensors with different cell constants exist.
Case Study:
Still, if you have doubts, then consider this case:
You have a line, and you need to measure the pressure in this line. Consider the following lines with pressure ranges as given below:
- A line having a pressure range of -100 mmWC to 100 mmWC
- A line having a pressure of 0 mmWC to 10000 mmWC
- Line having a pressure of 0 kg/cm2 to 1.5 kg/cm2
- Line having a pressure of 0kg/cm2 to 10 kg/cm2
- Line having a pressure of 0kg/cm2 to 200 kg/cm2
Will you use the same pressure transmitter for all these lines with different pressures? Definitely, we will not be able to measure all these lines’ pressure with the same pressure transmitter. Because the pressure transmitter, which ranges from 0 kg/cm2 to 200 kg/cm2, will not be able to measure pressure in a few hundred mmWC accurately. Similarly, using a conductivity sensor with the same cell constant, we cannot measure the conductivities of different liquids with different values.
For liquids with very low conductivities, the conductivity sensor with a high value of cell constant will not produce accurate values because the distance between the plates will be a bit high. Similarly, the conductivity sensor with a low cell constant value for liquids with high conductivities cannot measure an accurate conductivity value.
You can use the below-mentioned conductivity sensor with cell constants for the mentioned liquids.