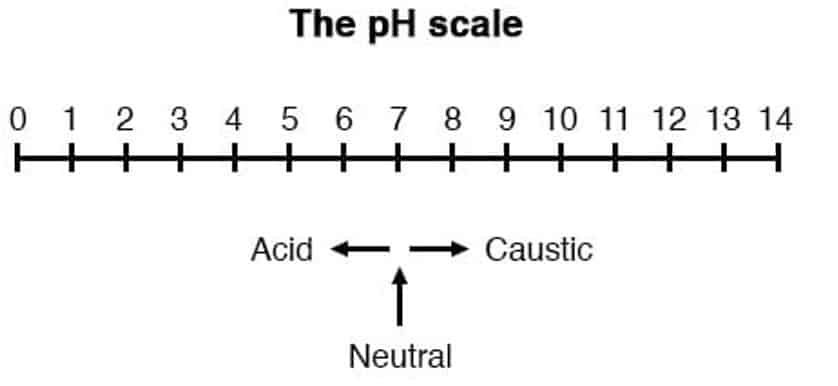
pH value is the potential of hydrogen in any given solution. The scale of pH is in the range of of 0 to 14. Based on pH value, we can categorize any solution as an acid or base or neutral solution.
If pH is between 0 to 7, then the solution is acid. The pH value nearer to 0 shows strong acid solution and values nearer to 7 shows weaker acids. The pH value of the neutral solution is 7. If the pH is between 7 to 14, then the solution is basic. The PH values near to 7 shows weaker base solution, and values nearer to 14 shows stronger base solution.
Calculation of pH value
The pH is the potential of hydrogen in any given solution. In other words, pH is the concentration of hydrogen ions in any given solution.
The formula of pH value is;
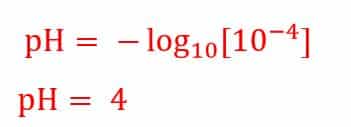
Similarly if the solution is having hydrogen ion concentration of 10-2 mol/l, then the pH value will be 2.
pH Measurement Procedure
- The manual method we use to know the pH of any given solution is using Litmus paper.
- A Litmus paper is a type of pH paper made by treating paper with natural dyes extracted from lichens.
- The Litmus paper pH test is done by placing small drops of the sample on colored paper.
- Litmus paper is usually red or blue. Red paper turns blue when the pH becomes alkaline, and blue paper turns red when the pH becomes acidic.
- Litmus paper is for only one time use. The Litmus paper just show the approximate value of the pH of the given solution.
- The pH measurement using litmus paper has inaccuracy. It is not possible to do continuous pH measurement because this method requires manual intervention.
- Also one major limitation of this litmus paper is that it turns white when exposed to Chlorine gas. This happens because of the bleaching effect.
- Other method of the pH measurement is using pH electrodes which is a type of electronic sensor. The sensor generates EMF based on the concentration of the hydrogen ion in the solution
- This method has 1 measuring electrode and 1 reference electrode which generate a potential difference when immersed in any given solution.
This potential difference is given by Nernst Equation.
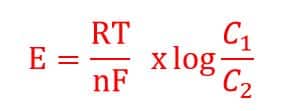
Where,
E = potential difference (mV)
R = gas constant (8.31439 J x mol-1 x K-1)
F = Faraday constant (96495.7 °C x mol-1)
T = absolute temperature in Kelvin (K)
n = charge number of the measured ion (in this case nH = 1)
C1 = active Hydrogen ion concentration in solution(our main solution) C1
C2 = active Hydrogen ion concentration in solution(filled in reference electrode) C2
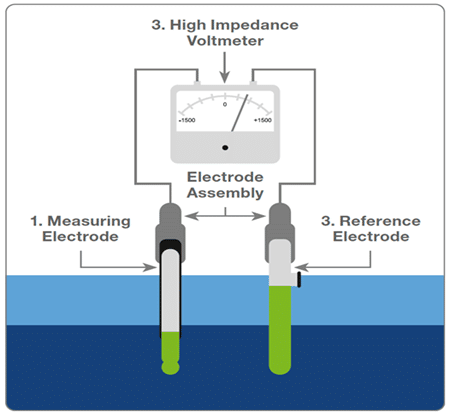
PH Electrodes
The below shown image is of combination electrode which has 2 different electrodes.

- Other type of pH electrode available is the single cell pH electrode or commonly known glass electrode.
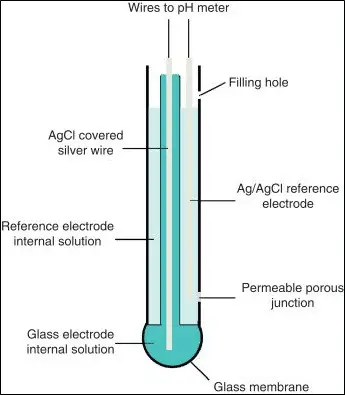
- The electrode consists of a pH-sensitive glass membrane and an internal Ag/AgCl reference electrode in 0.1 M HCl solution.
- The sample reference electrode is an Ag/AgCl electrode in the KCl solution (which is saturated with KCl or contains a fixed concentration of KCl.
- The porous wick acts as a salt bridge between the sample and the reference electrode.
How to increase performance and lifetime of pH electrode?
- To maximize the performance and life of your pH electrode, it is important to clean and calibrate the electrode regularly.
- If your electrode is not in use, the electrode should be stored in the recommended pH storage solution.
- The reference joint must not be dry.
- If your electrode exhibits slow settling times, erratic readings, or difficulty in calibration, it may be time to clean the electrode.
- The best cleaning solutions are those that are more selective in dealing with contamination (eg fats and oils should be washed with a non-ionic surfactant solution or methanol). After the cleaning solution, the electrode should be rinsed with deionized water and stored in a solution of the correct pH.
- If the electrode is reusable, the electrode must be filled with an appropriate electrode filling solution. Make sure there is enough electrolytes inside the electrode before use.
- Periodically calibrate the pH electrode.
- One very important factor which leads to inaccurate readings is the temperature compensation in pH electrodes. Generally all pH electrodes have a inbuilt RTD inside the sensor for temperature compensation because pH is a temperature dependent parameter. Therefore, temperature monitoring is a must for accurate measurement reading.
Output from pH electrode
- pH electrode gives output in millivolt.
- This millivolt us 59.16 millivolt/pH and this millivolt is given to pH transmitter. The transmitter converts this millivolt to appropriate signal understandable by the system(4 mA to 20 mA or FF signal or any other type of signal).
Read Next: