In this article, we will discuss the difference between cell and battery. Cell and battery are very important invention that has made our day-to-day life very easy. We use portable electronic equipment like cell phones, laptops, electronic watches, and many more, and these types of equipment can not function without the use of a cell or battery. In a nut cell, we can not imagine the world without the use of cell and battery.
Though the words cell and battery are used interchangeably, there is a big difference between them. The cell converts chemical energy into electrical energy. If we connect more cells in the series or parallel circuit, it forms a battery. Basically, the cell is the smallest unit of the battery. The battery is made by connecting the cells in the series and parallel combination as per requirements.
What is a Cell?
A cell is a single unit that stores chemical energy and converts it into electrical energy. The cell is an electrochemical device that uses chemical reactions to produce electricity. The important thing about the cell is that it is a portable device. Therefore, it is easy to carry. The symbol of cell is given below.
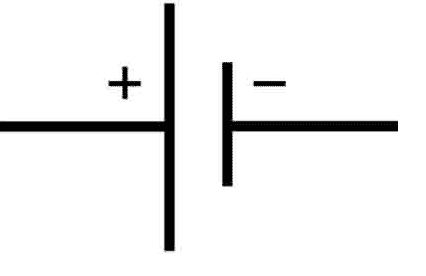
A cell has a chemical substance that reacts with the electrode and produces electricity. The cell has two electrodes-Cathode and Anode. The redox reaction takes place between the electrodes and electrolyte, and it leads to the flow of electric current in the external circuit. In a cell, the oxidation reaction takes place at the anode, whereas the reduction reaction happens at the cathode.
A cell maintains its stored energy till it is not in use. The cell provides a practical way of storing electrical energy because it does not lose its electrical energy if it is not in use. Therefore, the cells are widely used in household apparatus.
A cell is generally light in weight and compact in shape because it is a single unit. However, cells can not provide electrical energy for a longer period of time. A cell finds its widespread application in the apparatus, such as watches, radio, and remote control devices.
What is a Battery?
A battery is a group of cells that are connected in a series or parallel combination. A battery is an electrochemical device that converts chemical energy into electrical energy. The battery can store more electrical energy. A battery supplies DC energy to the load. The battery is used to supply power to equipment, such as mobile phones, flashlights, and cars. The symbol of battery is given below.
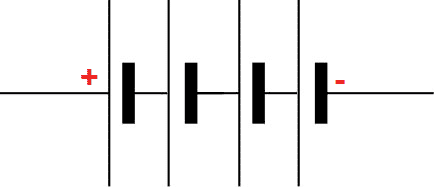
In above diagram, the four cells are connected in series to form a battery.
In every cell and battery, there are two electrodes- Anode and Cathode. The electrodes of the cell or battery have a certain distance and are submerged into the electrolytes. It is a must that there should be electrolytes between both electrodes for chemical reactions.
Types of Batteries
We can categorize the batteries into two types.
Primary Batteries
Primary batteries are those batteries that can not be reused after being used once. Therefore, we call it a single-use battery or a disposable battery. In primary batteries, the reactions during the discharging of the battery do not reverse, and it loses its charge after discharging.
Secondary Batteries
The secondary batteries can be recharged after their discharge. The charging of batteries is done by connecting the battery to a DC voltage source. During charging, the anode and cathode of the battery come in their original form.
Difference Between Cell and Battery
We can summarize the difference between cell and battery in the below table.
| Cell | Battery |
| A cell is a single-unit device that converts chemical energy into electrical energy. | A battery is a group of cells connected in series and parallel combinations. |
| A cell has a wet or dray type of electrolyte. | The battery may be of primary or secondary type. The primary battery can not be charged after discharge. The secondary battery can be charged after discharging. |
| Being a single unit, a cell is light in weight and compact. | A battery has several cells, therefore it is big and heavy in weight. |
| A cell can supply electricity for a shorter period of time. | A battery can supply electricity for a longer period of time. |
| A cell is used where there is a need for less electrical energy, such as watches, LED lights, etc. | A battery is used where there is a need for more electrical energy for a longer period of time. The batteries are used for inverters, automobiles, etc. |
| A cell is generally cheap. | Batteries are costly. |
Related Articles:
- Difference Between Primary Cell and Secondary Cell
- Why is the terminal voltage less than EMF during discharging of battery?
- Difference between Capacitor and Battery
- What is a single point load cell? its Working Principle
- What is Battery Acid? Its composition and Roles
- Battery Size Chart
- Internal Resistance of a Cell