Battery acid is a dilute solution of sulfuric acid (H₂SO₄) used in lead-acid batteries. Comprising 29%-32% sulfuric acid, it facilitates the flow of electrical current between the battery’s plates. This highly corrosive electrolyte is essential for generating electrical energy in vehicles and other applications. Proper handling and safety measures are crucial due to its hazardous nature.
A battery can not work without battery acid or electrolyte. The battery converts electrical energy into chemical energy during the charging process. The battery converts the chemical energy into electrical energy during discharging. We use the lead-acid batteries in motor vehicles, DG sets, and 110 volts DC source for electrical operated breakers. The electrolyte plays a very vital role in battery operation. In this article, we will learn about the composition of battery acid and its role in the battery charging and discharge process.

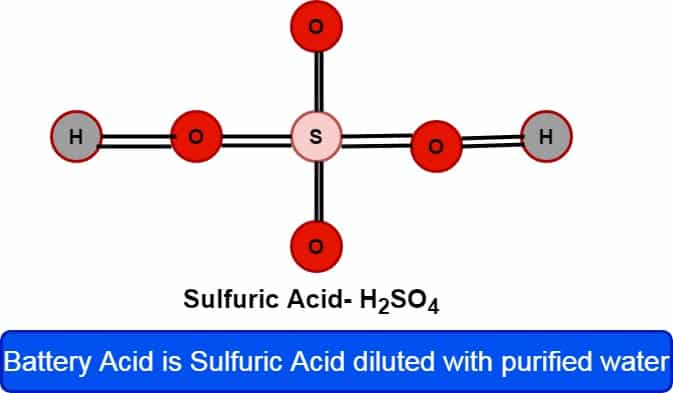
Battery Acid Composition
The battery acid is made of sulfuric acid (H2So4) diluted with purified water to get an overall concentration of around 29-32, a density of 1.25-1.28 kg/L, and a concentration of 4.2 mol/L.
The pH value of electrolytes is about 0.8, so we need to take utmost care when handling battery acid.
What Is Battery Acid?
- Battery acid is a common name for sulfuric acid (US) or sulphuric acid (UK).
- Sulfuric acid is a mineral acid with the chemical formula H2SO4.
- It is typically a mixture of sulfuric acid and water. The concentration of sulfuric acid in battery acid generally ranges from 29% to 32%.
- It is highly corrosive and can cause severe chemical burns on contact with skin or other materials.
- Battery acid reacts with metals, bases, and organic materials. Therefore, it is stored in glass or other nonreactive containers.
Why do batteries contain Acid?
The battery converts-
- Electrical energy into chemical energy during the charging process
- Chemical energy into electrical energy during the discharging process
The electrolyte is the medium that helps in converting electrical energy into chemical during charging and chemical energy into electrical during discharging. Thus, without battery acid, the functioning of the battery is impossible.
Construction of Battery
A lead-acid battery consists of two lead plates separated by an electrolyte. The positive plate has lead peroxide (PbO2), and the negative plate has lead (Pb). Diluted sulfuric acid remains as an electrolyte between the plates. The other part of the battery is the separator. The separators are the insulating material that keeps the battery plates apart to avoid touching them. The battery is rechargeable through charging and discharging chemical reactions.
Role of Battery Acid in Battery Charging & discharging
Electrons move from the negatively charged lead plate to the positively charged plate during the discharging of the battery.
The negative plate reaction is:
Pb(s) + HSO4-(aq) → PbSO4(s) + H+(aq) + 2 e-
The positive plate reaction is:
PbO2(s) + HSO4- + 3H+(aq) + 2 e- → PbSO4(s) + 2 H2O(l)
The combined overall chemical reaction is ;
Pb(s) + PbO2(s) + 2 H2SO4(aq) → 2 PbSO4(s) + 2 H2O(l)
The battery charging and discharge equations are given by

From the above equations, it is clear that the electrochemical reactions are not possible without electrolytes.
What happens during the Battery Charging and Discharge process?
When the battery is fully charged, the negative plate is lead(Pb), the electrolyte is concentrated sulfuric acid, and the positive plate is lead dioxide(Pb02). Under overcharging conditions, water electrolysis produces hydrogen and oxygen gas. Some types of batteries allow water addition to make up for the loss.
After battery discharging, the reverse reaction forms lead sulfate on both plates. The fully discharged battery cause formation of lead sulfate on both the plates separated by water. At this stage, the battery is completely dead and can’t recover or be charged again. Thus deep discharge of the battery causes permanent damage to the battery.
Never overcharge and deep discharge the battery.
Battery Acid Properties
Battery acid, which is typically diluted sulfuric acid (H₂SO₄), has several important properties:
- Concentration: In lead-acid batteries, the concentration of sulfuric acid usually ranges from 29% to 32%.
- Corrosive Nature: It is highly corrosive and can cause severe chemical burns. It can also corrode metals and other materials.
- Density: The density of battery acid is typically around 1.25 to 1.28 g/cm³, depending on its concentration.
- Boiling and Melting Points: Sulfuric acid has a boiling point of about 337°C (639°F) and a melting point of 10°C (50°F) in its pure form. However, these values can change when the acid is diluted.
- Viscosity: Sulfuric acid has a relatively high viscosity, which decreases with dilution.
- Conductivity: It is a good conductor of electricity when in an aqueous solution, which is why it is used in batteries.
- Reactivity: Sulfuric acid is highly reactive, especially with organic materials, metals, and bases. It can cause explosive reactions with certain chemicals, such as water (especially if water is added to the acid), leading to splattering and rapid boiling.
- Hygroscopic Nature: Sulfuric acid is highly hygroscopic, meaning it can absorb water from the air.
- Dehydrating Agent: It has strong dehydrating properties and can remove water from other substances, often charring organic materials by removing hydrogen and oxygen atoms as water.
Safety Measures While Handling Battery Acid
Proper handling and safety measures for battery acid are crucial due to its highly corrosive nature and potential hazards. Here are the essential steps to ensure safe handling:
Personal Protective Equipment (PPE)
When working with battery acid, it is vital to wear appropriate personal protective equipment. Acid-resistant gloves protect your hands, while safety goggles or a face shield protect your eyes from splashes. Wearing acid-resistant clothing, such as a lab coat or apron, is necessary to shield your skin. Additionally, closed-toe, acid-resistant shoes or boots provide protection for your feet.
Work Area Preparation
Prepare the work area by ensuring it is well-ventilated to avoid inhaling any fumes. An eyewash station and safety shower should be nearby in case of accidental exposure. Keep a spill kit with neutralizing agents and absorbent materials readily available to handle any spills promptly.
Handling Procedures
When transporting battery acid, use appropriate containers and carriers designed for corrosive liquids, ensuring they are securely sealed. Pouring battery acid should be done slowly and carefully to prevent splashes. Always add acid to water, never water to acid, to avoid violent reactions. Use glass or acid-resistant plastic containers for mixing, and avoid using metal containers as they can corrode.
Storage
Store battery acid in labeled, corrosion-resistant containers with tight-fitting lids. The storage location should be cool, dry, and well-ventilated, away from direct sunlight and incompatible materials such as bases and organic substances. Ensure the storage area is secure to prevent unauthorized access.
Disposal
Dispose of battery acid properly by neutralizing small amounts of spilled acid with a suitable neutralizing agent, such as baking soda, before disposal. For large amounts, dispose of the acid as hazardous waste, following local regulations and guidelines.
First Aid
In case of skin contact with battery acid, rinse the affected area with plenty of water for at least 15 minutes and seek medical attention. If the acid comes into contact with the eyes, flush them with water for at least 15 minutes, keeping the eyelids open, and seek immediate medical attention. If inhalation occurs, move to fresh air immediately and seek medical attention if symptoms persist. In the event of ingestion, do not induce vomiting; rinse the mouth with water and seek immediate medical attention.
By adhering to these comprehensive safety measures, you can minimize the risks associated with handling battery acid and maintain a safe working environment.
Good to Know-Sulfuric Acid Names and Concentrations
Sulfuric acid, commonly referred to as “battery acid” when at a specific concentration, has several names that typically reflect its concentration and usage:
- Dilute Sulfuric Acid: This term is used for sulfuric acid with a concentration less than 29% or 4.2 mol/L.
- Battery Acid: This is sulfuric acid with a concentration of 29-32% or 4.2-5.0 mol/L, commonly found in lead-acid batteries.
- Chamber Acid or Fertilizer Acid: Sulfuric acid at a concentration of 62-70% or 9.2-11.5 mol/L, produced using the lead chamber process.
- Tower Acid or Glover Acid: This term refers to sulfuric acid with a concentration of 78-80% or 13.5-14.0 mol/L, which is recovered from the bottom of the Glover tower.
- 66 °Bé (“66-degree Baumé”) Acid: This is sulfuric acid with a concentration of 93.2% or 17.4 mol/L, named for its density as measured by a hydrometer.
- Concentrated Sulfuric Acid: Refers to sulfuric acid with a concentration of 98.3% or 18.4 mol/L. While nearly 100% sulfuric acid can be made, it typically stabilizes at 98.3% due to the loss of SO₃ near its boiling point.
Frequently Asked Questions (FAQs) on Battery Acid
1. What is battery acid?
Battery acid is typically a diluted form of sulfuric acid (H₂SO₄) used in lead-acid batteries. It is highly corrosive and can cause severe burns or injury upon contact with skin or other materials.
2. How dangerous is battery acid?
Battery acid is extremely dangerous. It can cause chemical burns, damage to eyes, respiratory issues if inhaled, and severe damage to materials such as metal, fabric, and other surfaces. Immediate medical attention is required if it comes into contact with skin or eyes.
3. What should I do if I come into contact with battery acid?
- Skin Contact: Rinse the affected area with plenty of water for at least 15 minutes and seek medical attention.
- Eye Contact: Flush the eyes with water for at least 15 minutes and seek immediate medical help.
- Inhalation: Move to fresh air and seek medical attention if you experience difficulty breathing or other symptoms.
4. How should battery acid spills be cleaned up?
- Small Spills: Neutralize with a baking soda (sodium bicarbonate) solution, then clean with water.
- Large Spills: Evacuate the area and call emergency services or a professional hazmat team. Always wear protective gear when dealing with battery acid spills.
5. Can battery acid be recycled or disposed of safely?
Yes, battery acid should be handled and disposed of by professionals. Lead-acid batteries can be recycled at designated facilities that handle hazardous materials.
6. How can I protect myself when handling battery acid?
- Wear appropriate personal protective equipment (PPE) such as gloves, goggles, and acid-resistant clothing.
- Work in a well-ventilated area.
- Have neutralizing agents like baking soda and plenty of water nearby in case of spills.
7. What are the common uses of battery acid?
Battery acid is mainly used in lead-acid batteries, which are commonly found in vehicles, backup power systems, and industrial equipment.
8. Can battery acid corrode other materials?
Yes, battery acid is highly corrosive and can damage metals, concrete, fabrics, and many other materials. It is important to handle it with care and store it in appropriate containers.
9. What are the symptoms of battery acid poisoning?
- Inhalation: Coughing, difficulty breathing, throat irritation.
- Ingestion: Severe abdominal pain, vomiting, diarrhea.
- Skin/Eye Contact: Severe burns, pain, redness, swelling, vision loss if in eyes.
10. Is there a way to neutralize battery acid?
Yes, battery acid can be neutralized with a solution of baking soda and water. The reaction will produce carbon dioxide gas, so it should be done in a well-ventilated area.