Coulomb is the SI unit of electric charge. One coulomb is equal to the amount of electric charge transferred by a current of one ampere in one second. Coulomb is the SI unit of electric charge.
What is Coulomb?
The coulomb is defined as the quantity of electric charge transported in one second when the current flow is one ampere. Coulomb is named for French physicist Charles -Augustin de Coulomb. One Coulomb is approximately equivalent to 6.24 × 1018 electrons. The charge of one electron is equal to 1.602176634 × 10−19 C. Number of charges in one coulomb is;
One coulomb is the charge of approximately 6241509074460762607.776 elementary charges,
Formula for Finding charges in Coulombs
The following formula shows the relationship between coulomb and ampere.
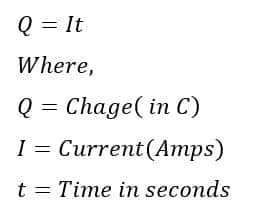
For example, if 3 amperes current flow in a circuit for 3 seconds, then the charges transported in the circuit are;
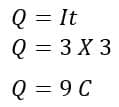
Symbol of Coulomb?
Coulomb is denoted by the letter C. It is the SI unit of electric charge.
Mathematically, coulomb is;
1 Coulomb = 1 ampere X 1 second
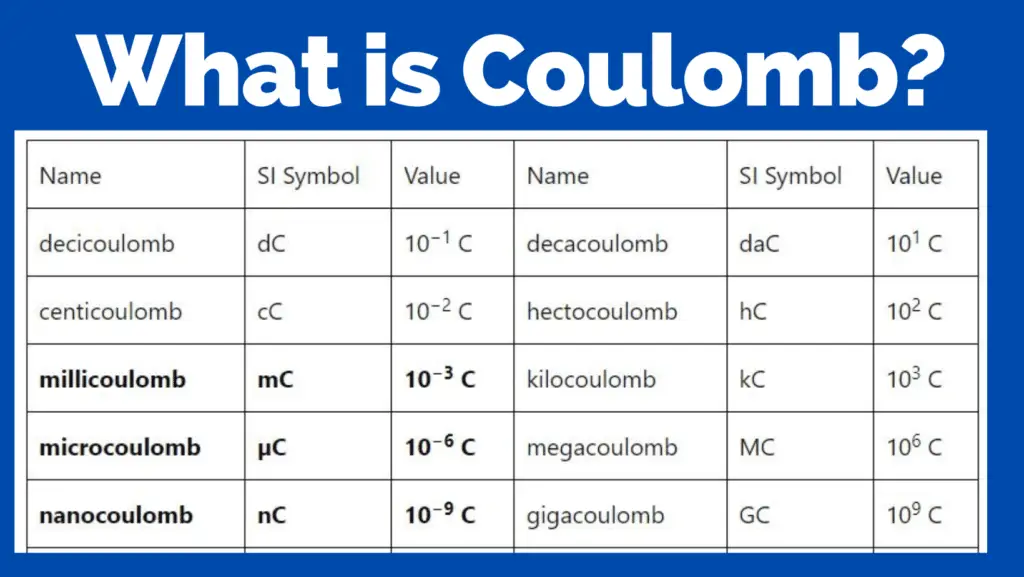
Other Units of Coulomb
We can get the other units of coulomb by adding a prefix a multiplies it by a power of 10.
| Name | SI Symbol | Value | Name | SI Symbol | Value |
| decicoulomb | dC | 10−1 C | decacoulomb | daC | 101 C |
| centicoulomb | cC | 10−2 C | hectocoulomb | hC | 102 C |
| millicoulomb | mC | 10−3 C | kilocoulomb | kC | 103 C |
| microcoulomb | µC | 10−6 C | megacoulomb | MC | 106 C |
| nanocoulomb | nC | 10−9 C | gigacoulomb | GC | 109 C |
| picocoulomb | pC | 10−12 C | teracoulomb | TC | 1012 C |
| femtocoulomb | fC | 10−15 C | petacoulomb | PC | 1015 C |
| attocoulomb | aC | 10−18 C | exacoulomb | EC | 1018 C |
| zeptocoulomb | zC | 10−21 C | zettacoulomb | ZC | 1021 C |
| yoctocoulomb | yC | 10−24 C | yottacoulomb | YC | 1024 C |
| rontocoulomb | rC | 10−27 C | ronnacoulomb | RC | 1027 C |
| quectocoulomb | qC | 10−30 C | quettacoulomb | QC | 1030 C |
Definition of One Coulomb on the basis of coulomb’s law
Coulomb’s law states that the electrical force between two charged particles is directly proportional to the product of the charge quantity and inversely proportional to the square of the separation distance between the charged particles.
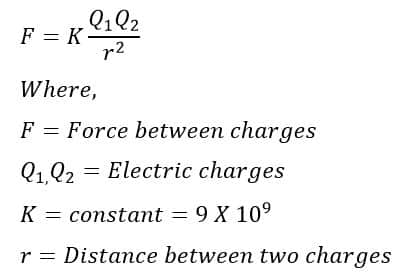
Now, to find the coulomb charge, we consider the followings.
Distance between the charges = 1 Meter
The force between charges = 9 X 109 N
Value of constant = 9 X 109
Q1=Q2 =Q ( Two equal charges)
Putting the above values in the coulomb’s law formula, we get
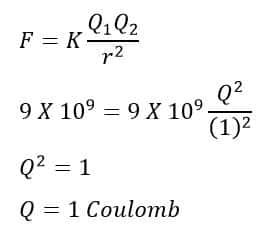
From the above expression, it is clear that the one coulomb is the quantity of charge that experiences a repulsive force F=9×109 N when placed at a distance of 1 meter in vacuum or air from an equal charge.