In this guide, you’ll discover everything about Coulomb’s Law — one of the cornerstones of electrostatics. We’ll cover its statement, formula, derivation, limitations, and applications in a clear, simple way so you can understand how electric charges interact in real life.
What is Coulomb’s Law?
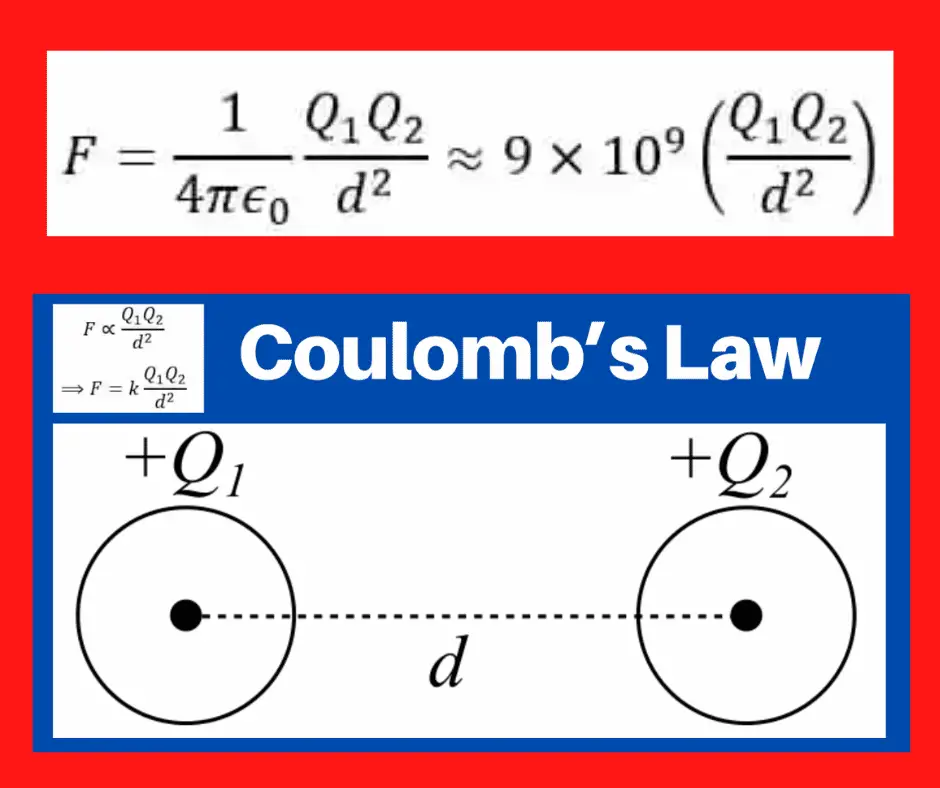
Coulomb’s Law describes how electric charges exert force on each other.
Proposed by Charles Augustin de Coulomb in 1785, the law states that the electrostatic force between two stationary point charges is:
- Directly proportional to the product of their charges.
- Inversely proportional to the square of the distance between them.
Because the force decreases rapidly as the distance increases, it is also called the inverse square law in electrostatics.
Coulomb’s experiments with a torsion balance laid the foundation for understanding electric and magnetic fields — concepts that later led to Maxwell’s equations.
Statement of Coulomb’s Law
”The electrostatic force between two point charges is directly proportional to the product of their magnitudes and inversely proportional to the square of the distance separating them.”
Mathematically,
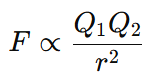
When we include the proportionality constant k, the equation becomes:
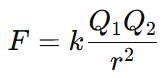
Coulomb’s Law Formula and Derivation
Imagine two point charges Q₁ and Q₂ separated by a distance d.
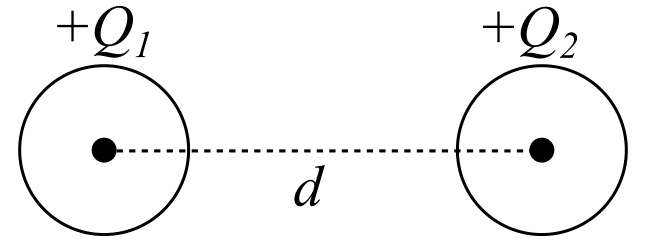
According to Coulomb’s Law, the force between them is,
- The electrostatic force is directly proportional to the product of charges
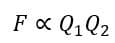
- The electrostatic force is inversely proportional to the square of the distance between the centers of the charged bodies, i.e.
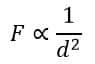
Combining these two equations, we get,
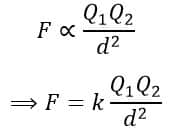
Where k is a constant of proportionality whose value depends on the medium in which the bodies are placed.
In the SI system, the value of k is given by,
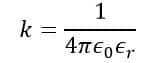
Where,
- ε0=8.85×10−12 C2/N⋅m2=8.85×10−12C2/N⋅m2 → permittivity of free space
- εr → relative permittivity of the medium
Therefore, the formula of the electrostatic force becomes,
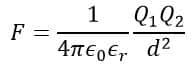
When bodies are separated from each other in a vacuum, then εr=1, thus,
Key Notes:
- If both charges are like charges (positive–positive or negative–negative), the force is repulsive.
- If the charges are unlike (positive–negative), the force is attractive.
- The force always acts along the line joining the two charges.
Coulomb’s Law Formula in Vector Form
If two point charges Q₁ and Q₂ are separated by a distance d, the force on Q2 due to Q1 is:
Where:
- F21 → Force on charge Q2 due to Q1
- ε0→ Permittivity of free space (8.854 × 10⁻¹² C²/N·m²)
- d → Distance between the charges
- d^21 → Unit vector pointing from Q1 to Q2
Key Points:
- Force magnitude: F=1/4πε0(Q1Q2/d2)
- Direction: Along the line joining the charges
- Repulsive if charges are alike, attractive if charges are opposite
Generalized Vector Form:
Limitations of Coulomb’s Law
Coulomb’s Law is accurate under specific conditions. Below are its major limitations-
- Applicable only for point charges: The law assumes charges are point-sized; it fails for large or irregularly shaped charged bodies.
- Valid only for stationary charges: It cannot be applied to moving charges, since magnetic effects aren’t considered.
- Fails in non-uniform media: Coulomb’s Law assumes a uniform medium; it’s inaccurate in materials where permittivity changes.
- Not accurate for atomic or planetary distances: At very small or very large scales, other forces dominate.
In simple terms, Coulomb’s Law is reliable only for small, stationary point charges placed in a vacuum or uniform dielectric medium.
Applications of Coulomb’s Law
Coulomb’s Law is widely used in physics, engineering, and electrical systems. Here are its key applications:
- To calculate electrostatic force:
It determines the magnitude and direction of force between two point charges. - To measure the distance between charges:
Using known charge values and measured force, distance between charges can be calculated. - To determine net electrostatic force on a charge:
When multiple charges act on a single charge, the resultant force can be computed using vector addition based on Coulomb’s Law. - In electric field and potential calculations:
The law serves as the foundation for deriving the electric field intensity and potential due to a charge distribution. - In designing capacitors and insulating materials:
Engineers use Coulomb’s Law to understand how dielectric materials affect electric fields and energy storage.
Example Problems
Example 1 – Calculate the electrostatic force between two charges 7 × 10-8 C and 2 × 10-7 C. These two charges are separated by a distance of 15 cm in free space.
Solution – Given data,
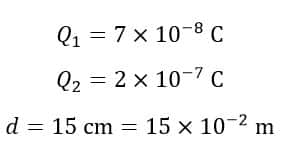
According to Coulomb’s law, we have,
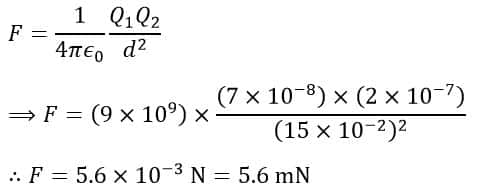
Example 2 – Two spheres are separated by a distance of 4 cm in air. The electrostatic charge on one sphere is 5 × 10-9 C and the electric force of attraction of 5 mN is acting between the spheres. Determine the electric charge on the second sphere.
Solution – Given data,
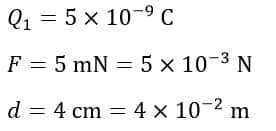
According to Coulomb’s law, we have,
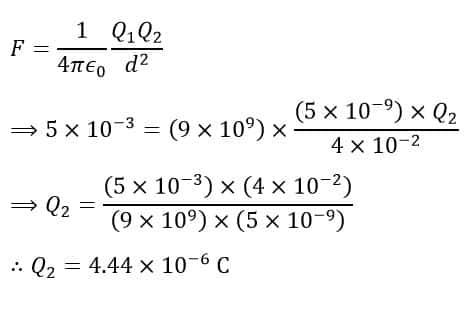
Conclusion
Coulomb’s Law provides the basic understanding of how electric charges interact.
Although it has some limitations, it remains a fundamental principle for studying electrostatics, designing capacitors, and calculating electric fields and potentials.
By mastering this law, you build the foundation to understand Gauss’s Law, Electric Field, and Electrostatic Potential — the next steps in learning electricity and magnetism.
Frequently Asked Questions (FAQs) on Coulomb’s Law
Coulomb’s Law states that the electrostatic force between two charged bodies is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. In short, larger charges or shorter distances create stronger forces.
Coulomb’s Law was discovered by Charles-Augustin de Coulomb in 1785 using a torsion balance experiment to measure the force between electric charges.
– It is valid only for point charges at rest.
– It cannot be applied to large or irregularly shaped bodies.
– It does not consider medium non-uniformity or relativistic effects.
Coulomb’s Law helps to:
– Calculate the electrostatic force between charges.
– Find the distance or charge magnitude when the force is known.
– Analyze electric fields and potential in electrostatics.
Related Articles:
- Ohm’s Law-Satatement, Formula, Solved Examples
- Limitations of Ohm’s Law
- Faraday’s Laws of Electromagnetic Induction
- Biot Savart Law: Statement, Derivation, Importance & Applications
- Fleming’s Left Hand Rule and Right Hand Rule
- Faraday’s Laws of Electrolysis – First and Second Laws
- Coulomb’s Law in Vector Form- Formula, Derivation, Applications