This article describes oxygen measurement in flue gas. There are two types of oxygen analyzer-Paramagnetic and Zirconia– used to measure oxygen in flue gas.
What is flue gas?
After the combustion process in the boiler, flue gas contains a gas mixture with polluting nature, and solid particles exhaust into the environment through the stack. Emissions are important to the proper functioning of almost every industrial process that burns fuel. This is true from clean-burning natural gas to coal furnaces.
Flue gas oxygen content is one of the important parameters for boiler operation, it indicates how efficient boiler operation is.
Purpose of O2 measurement in flue gas
The optimum ratio of fuel and combustion air requirement is found from oxygen measurement in the flue gas. An oxygen analyzer is a perfect instrument to monitor the amount of excess air and heat loss. To keep high combustion efficiency levels, oxygen and fuels must be measured.
Oxygen measurements in the flue gas are generally used to set the fuel-air ratio in combustion. Flue gas analyzers allow these measurements to be made ensuring efficient boiler operation.
What are the constituents of flue gas?
The efficiency of the boiler boosts up as a result of an understanding of the constituents of flue gas and correct analysis of their proportions. Excess air results in oxygen that are not consumed carrying away heat with flue gas. Whereas too little air results in poor mixing with fuel leading to the emission of heavy gas into the stake.
In the boiler combustion process, a variety of gases and products are generated. The most important are CO (Carbon Monoxide), CO2 (Carbon Dioxide), O2 (Oxygen), unburned hydrocarbons, solid particles, Nitrogen, Water, and under certain conditions NOx (Nitrogen oxide) are among them.
When the fuel and oxygen are completely burned in the combustion process, produce only CO2 (Carbon dioxide) and H2O (water).
There are two kinds of technologies for the measurement of O2 in flue gases in the stack. Following are the type of instruments that will allow us to perform flue gas analysis.
1. Paramagnetic Oxygen analyzer
2. Zirconia Oxygen measurement
Paramagnetic O2 analyzer
Oxygen is paramagnetic in nature. Hence, Oxygen has the property of getting attracted to a strong magnetic field. Measurement by paramagnetic is purely physical. It ensures that there, no waste is generated. Measuring cells can live for a long period. However, the performance of the cell can be degraded with dirt, dust, corrosives, or solvents. Oxygen is normally measured between 0.05% to 100%.
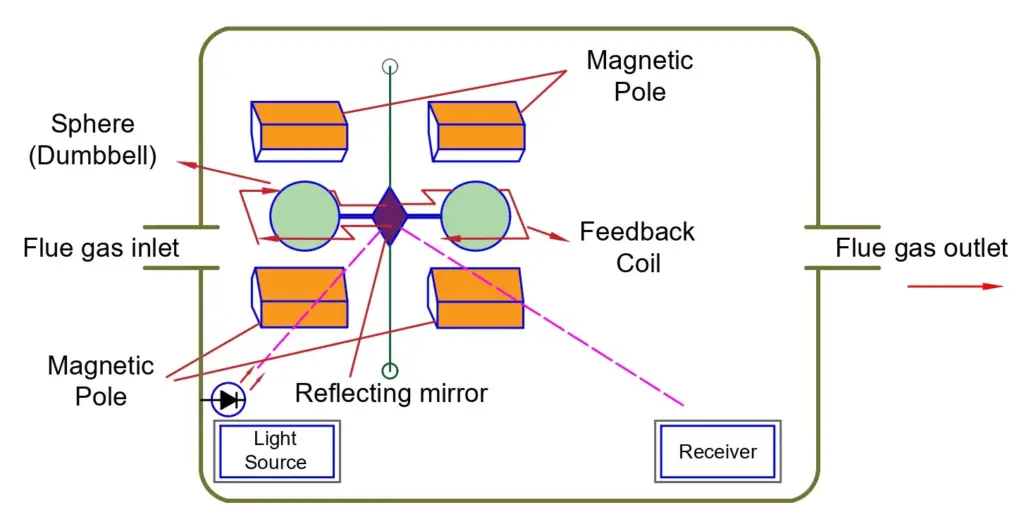
The paramagnetic oxygen analyzer measures oxygen in gases or gas mixtures at any level between 0-100 percent.
Working Principle of Paramagnetic Analyzer
The working principle of the paramagnetic sensor is the paramagnetic susceptibility of the oxygen molecule. It is a physical property that distinguishes oxygen from most other gases.
The sensor part comprises two nitrogen-filled glass spheres mounted on a rotating suspension. The total assembly is suspended in a strong magnetic field. Oxygen from the surrounding gas is attracted to the magnetic field, causing a force on the glass spheres. Depending on the oxygen content of the surrounding gases, torque forces are applied to the suspension.
Operation of Paramagnetic analyzer
In the first step of measurement, nitrogen is passed through the suspension assembly. The assembly will be in a balanced state with the system output as “null”. The zero position is detected by a photosensor that receives light reflected from a mirror attached to the suspension assembly. The output from the photosensor is returned to a coil around the suspension assembly.
When oxygen is introduced into the cell, the torque acting on the suspension assembly is balanced by a reset torque due to the return current in the coil. The feedback current is directly proportional to the volumetric magnetic susceptibility of the sample gas. The partial pressure of oxygen in the sample, therefore, is the current.
It provides an accurate measurement of the oxygen concentration in the gas mixture.
Benefits of Paramagnetic analyzer
- Maintenance is minimum, and the sensor is long-lasting.
- Response time is fast, with good accuracy.
- Highly sensitive measures even low levels of oxygen.
Zirconia Oxygen Analyzer
The Zirconium Oxygen Analyzer is suitable for measuring ppm to % oxygen levels in a gas mixture. The zirconia cell is an electrochemical galvanic cell that uses a high–temperature ceramic sensor containing stabilized zirconia.
Inside an instrument, the zirconia cell is mounted in a temperature-controlled furnace with the necessary electronics to process the signal from the detection cell. Measurements are normally displayed directly via a digital display as the oxygen concentration is in the range of 0.01 ppm to 100%.
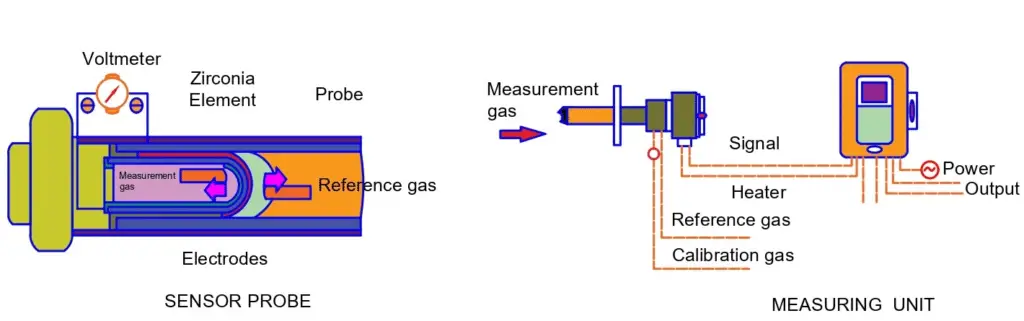
Construction of Zirconia cell
The zirconium cell is a high-temperature ceramic sensor and is an electromechanical galvanic cell. It is made up of two electrically conductive chemically inert electrodes. As shown in the above schematic figure, they are fixed to both sides of the solid electrolyte.
The ceramic tube (stabilized zirconium oxide) is completely gas-tight and conducts electricity using oxygen ions when functioning at operating temperatures. Sensors of this type must be heated to above 450° C before it becomes electrolyte conductor. Nernst equation postulates the potential difference across the cell.
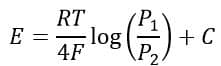
Where,
E = Potential difference
R = Gas constant
T = Absolute temperature
F = Faradays constant
C = Cell constant of sensor.
P1 and P2 are the partial pressures of oxygen on either side of the zirconium tube.
If one electrode has a known partial pressure and the temperature of the sensor is controlled, the potential difference between the two electrodes can be used to determine the unknown partial pressure.
Operating Principle of Zirconia Oxygen Analyzer
The zirconia cell is made of the yttrium oxide stabilized zirconium oxide as a ceramic with porous platinum electrodes.
Molecular oxygen ionizes on porous platinum electrodes. The platinum electrode on each side of the cell provides a catalytic surface for changing oxygen molecules into oxygen ions and oxygen ions into oxygen molecules.
When the oxygen molecules gain electrons, they become ions that enter the electrolyte on the high-concentration side of the cell. At the same time, on the other electrode, oxygen ions lose electrons and are released as oxygen molecules.
The oxygen content of these gases, and therefore the partial pressures of oxygen, is different. Each electrode produces oxygen ions at a different rate and introduces them into the zirconium oxide electrode at a different rate. As zirconium oxide allows oxygen ions to move, the number of oxygen ions moving in each direction will depend on the rate at which oxygen ions ionize and enter the electrolyte. It involves vacancies in the zirconium oxide lattice through doping with yttrium oxide, which is a complex mechanism.
Based on the partial pressures o of oxygen at the two electrodes, the migration of oxygen ions through the electrolyte results in a net flux of ions in one direction.
The zirconium oxide analyzer uses air as a reference gas. In which a constant oxygen concentration of 20.9% is maintained. Temperature control of the furnace is maintained at 650Deg.C.
Zirconium oxide analyzer electronically calculates the oxygen partial pressure, and therefore the oxygen concentration, of a gas sample with a concentration unknown oxygen intake. A potential difference “E” is measured across the electrodes of the zirconium cell.
Benefits of Zirconia analyzer
- Direct measurement of O2, no need to extract and condition the sample.
- Zirconia cells are used to measure O2 in the high-temperature flue gas.
- O2 measurement in all types of fuels.
- Faster response
Read Next: