In this article, we will discuss the dissolved oxygen analyzer working principle. A dissolved oxygen sensor monitors the concentration of dissolved oxygen in the water.
What is Dissolved Oxygen?
Dissolved oxygen is the amount of oxygen that is dissolved in the water. The dissolved oxygen is represented by mg/L. We measure dissolved oxygen because oxygen is an essential element for the respiration of underwater creatures, and dissolved oxygen also acts as a chemical oxidizer. The solubility of oxygen in any given water is significantly affected by the water’s temperature, salinity, and barometric pressure.
Measurement of Dissolved Oxygen
1. Electrochemical Dissolved Oxygen Sensors
Electrochemical dissolved oxygen sensors are sometimes referred to as amperometry or Clark-type sensors. Two types of electrochemical Dissolved Oxygen sensors are available: galvanic and polarographic. Polarograph Dissolved Oxygen Sensors can be divided into static and fast pulse sensors. The galvanic Dissolved Oxygen sensor and the polarograph Dissolved Oxygen sensor use two polarization electrodes (anode and cathode) in the electrolyte. A thin semipermeable membrane separates the electrode and electrolyte from the sample.
When measuring, dissolved oxygen diffuses through the membrane in proportion to the oxygen pressure in the water. Dissolved oxygen is then reduced and used at the cathode. This reaction produces a current that is directly proportional to the oxygen concentration. This current is always carried by the ions that are present in the electrolyte and flow to the anode from the cathode.
Galvanic Dissolved Oxygen Sensors
In galvanic dissolved oxygen sensors, the electrodes are made of dissimilar metals. Metals have different potentials based on their sequence of activity (how easily they donate or accept electrons). It self-polarizes when placed in an electrolytic solution due to the potential between dissimilar metals.
This self-polarization means that the galvanic oxygen sensor does not require warm-up time. The potential difference between the anode and cathode should be at least 0.5 volts to reduce oxygen without external potential.
The anode of the galvanic dissolved oxygen sensor is usually zinc, lead, or another active metal, and the cathode is silver or another precious metal. The electrolyte solution can be sodium hydroxide, sodium chloride, or another inert electrolyte. The electrochemical response of the galvanic oxygen sensor is very similar to that of the polarograph oxygen sensor but does not require a separate constant potential.
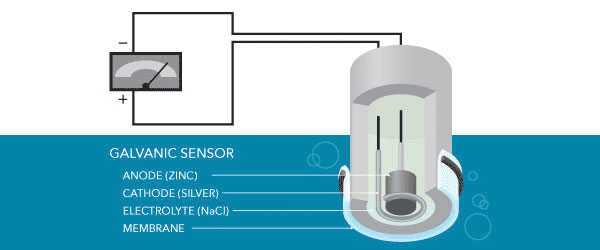
The different electrodes are self-polarized, and the electrons move internally from the anode to the cathode. The cathode remains inert, functions only to transfer electrons, and does not interfere with the reaction. This oxidizes the anode and reduces oxygen on the surface of the cathode. Due to this effect, a current starts flowing, which can be converted to a proportional 4 mA to 20 mA or other signal using a suitable transmitter.
Polarographic Dissolved Oxygen Sensors
Polarographic Dissolved Oxygen sensors are electrochemical sensors consisting of a silver anode and a noble metal cathode (gold, platinum, rarely silver, etc.) in a potassium chloride (KCl) solution. If the equipment is turned on, a 5 to 60-minute warm-up period is required to polarize the electrodes before calibration or measurement.
The electrodes are polarized by a constant voltage from the cathode to the anode (needed between 0.4V and 1.2V to reduce oxygen). When the electrons move in the opposite direction of the current, the anode is positively polarized, and the cathode is negatively polarized.
This polarization occurs when electrons move from the anode to the cathode through the internal circuitry of the wire. As oxygen diffuses through the membrane, the molecules of the cathode decrease, and the electrical signal increases.
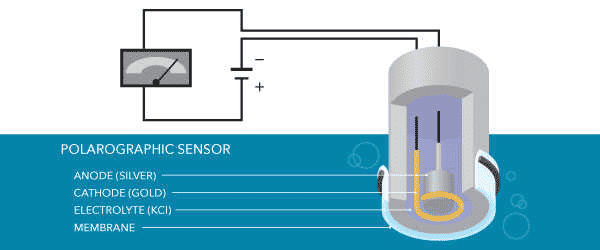
The polarization potential remains constant while the sensor detects the change in current caused by the reduction of dissolved oxygen. As more oxygen passes through the membrane and decreases, the current measured by the Polarograph Dissolved Oxygen sensor increases. A measurable current is produced and converted to a suitable signal using a transmitter.
2. Optical Sensors
An optical dissolved oxygen sensor measures the interaction of oxygen with a particular luminescent dye. When exposed to blue light, these dyes are excited (electrons acquire energy) and emit light when the electrons return to their normal energy state. In dissolved oxygen, oxygen molecules interact with the dye, limiting or altering the wavelength returned.
The measured effect is inversely proportional to the oxygen partial pressure. Some optical DO sensors are called fluorescent sensors, but the term is technically incorrect. These sensors emit blue light instead of ultraviolet light and are known as optical or luminescent DO sensors.
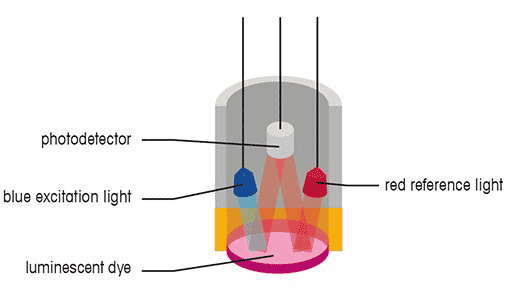
The optical dissolved oxygen sensor can measure either the intensity or the lifetime of the luminescence because oxygen affects both.
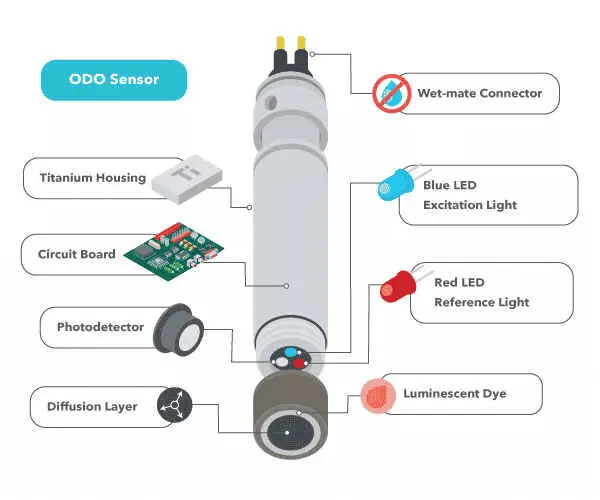
The optical DO sensor comprises a semipermeable membrane, sensor element, light-emitting diode (LED), and photodetector. The sensor element contains a sol-gel, xerogel, or fluorochrome immobilized on another matrix. This dye reacts when exposed to the blue light emitted by the LED.
Some sensors emit red light as a reference to ensure accuracy. This red light does not cause light emission but only reflects off the dye and returns. When exposed to blue light, the intensity and luminescence lifetime of the dye depends on the amount of dissolved oxygen in the water sample. As oxygen passes through the membrane, it interacts with the dye, limiting the intensity and lifetime of the luminescence.
The intensity or lifetime of the returned luminescence is measured by the photodetector and is used to calculate the dissolved oxygen concentration. A transmitter converts this into a suitable signal understandable by the system.