In this article, we will understand the two fundamental laws of electrolysis, which are known as Faraday’s laws of electrolysis. Electrolysis is an electrochemical process in which an electric current drives a non-spontaneous reaction. Faraday’s first and second laws are basically two statements that describe the electrolysis process mathematically.
What is Electrolysis?
An electrochemical process in which an electric current passes through an electrolyte solution, and a chemical reaction occurs in this solution, called electrolysis.
An arrangement to perform electrolysis is shown in the following figure.
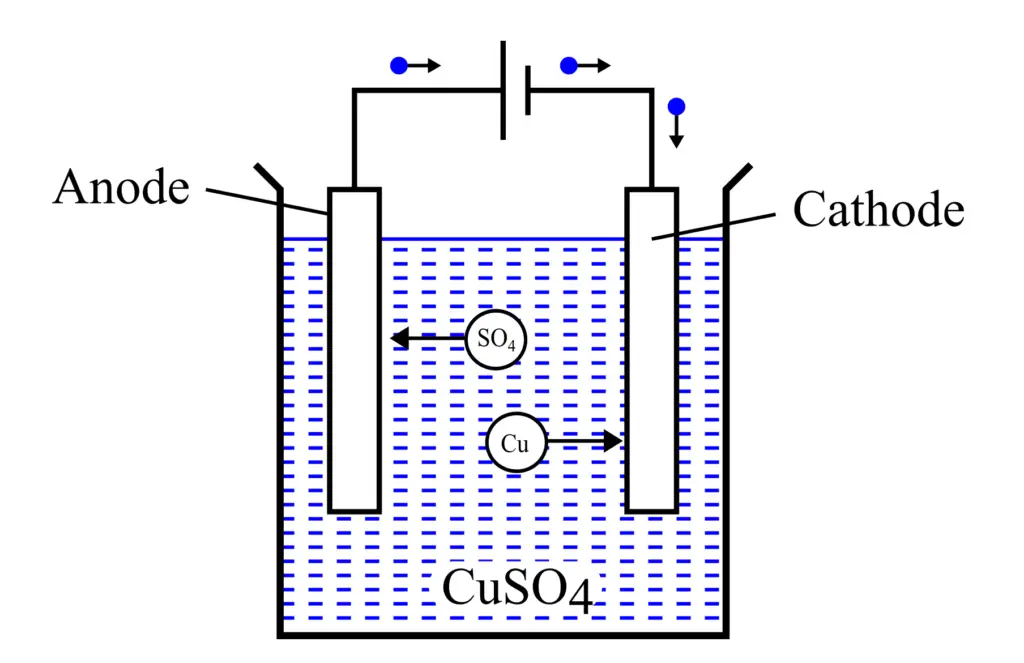
It is a copper voltameter. It consists of a positive electrode called the anode, a negative electrode called the cathode, and a water solution of copper sulfate (CuSO4).
When CuSO4 is dissolved in water, it breaks into its constituents, namely copper ions (Cu) and sulfate ions (SO4), where the copper ions bear a positive charge while the sulfate ions bear a negative charge.
When a direct current flows through the electrodes, the negatively charged sulfate ions move toward the anode, and the positively charged copper ions move toward the cathode. Hence, a chemical reaction occurs together with the flow of electric current.
This is how the process of electrolysis takes place. It is also important to note that electrolysis only occurs if a direct current or DC voltage is applied to the electrodes.
Electrolysis is widely used in several applications, such as water electrolysis, electroplating, electrorefining, and more.
After an introduction to electrolysis, let us discuss Fraday’s first and second laws of electrolysis.
Faraday’s First Law of Electrolysis
Faraday’s first law of electrolysis states that the mass of deposition or liberation of substance due to the flow of electric current through an electrolyte is directly proportional to the amount of electric charge that passes through it, i.e.,
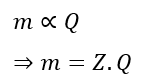
Where Z is a constant of proportionality called the electrochemical equivalent of the substance.
Since,
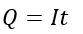
Where I is current, and t is time.
Therefore,
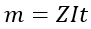
The electrochemical equivalent (Z) value is the same for an element and different for others.
Definition of Electrochemical Equivalent:
If charge Q is equal to 1 coulomb, then m = Z.
Thus, the electrochemical equivalent can be defined as the mass of the substance deposited on the electrode when 1 coulomb of charge passes through the electrolyte.
The electrochemical equivalent of an element or substance is measured in gm/C or kg/C.
Faraday’s Second Law of Electrolysis
This law specifies the quantitative relationship between the mass of the element deposited and the chemical equivalent weight of the element.
According to this law, the mass of an element or substance deposited during the process of electrolysis is proportional to the chemical equivalent weight of that element or substance, i.e.,
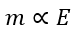
Where E is the chemical equivalent weight of an element or substance.
To understand the second law of electrolysis, let us perform an experiment.
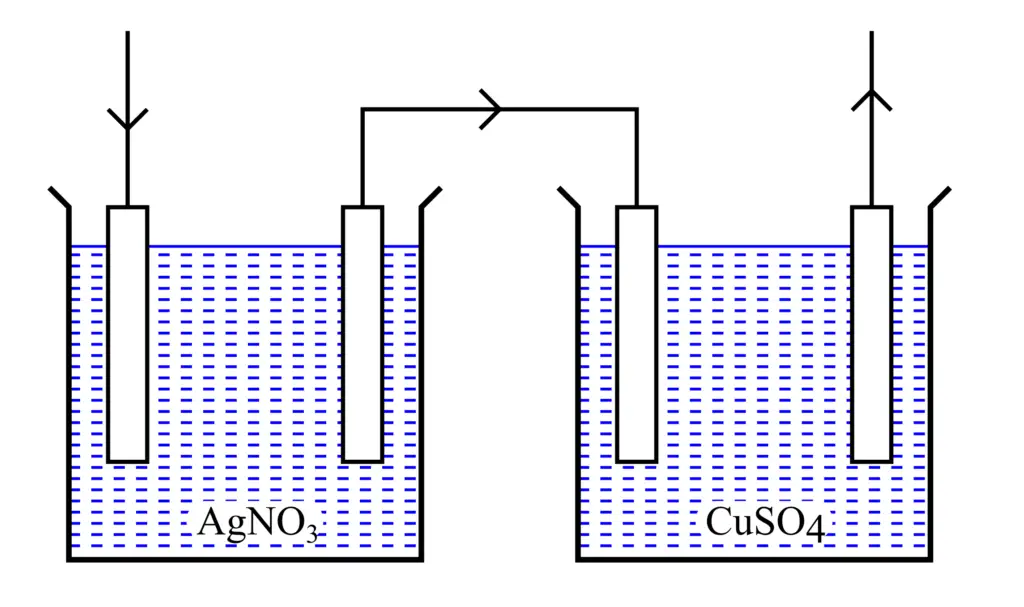
Consider two voltameters: one is a silver voltameter, and the other is a copper voltameter. These two voltameters are connected in series with the help of a conductor so that the same current flows through them.
When an electric current is passed through these two voltameters for the same amount of time, then we will find that the silver and copper deposited on the respective cathodes will be in the ratio of 108:32. Where 108 and 32 are the chemical equivalent weights of the silver and copper respectively.
Chemical Equivalent Weight:
For an element, the chemical equivalent is defined as the ratio of atomic weight of the element and its valency, i.e.,
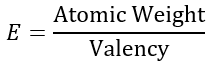
Relationship between E and Z
We can determine the relationship between an element’s electrochemical equivalent (Z) and the chemical equivalent weight (E).
For this, let us consider two substances that are deposited on cathodes, and their masses are m1 and m2. The ECEs of these two substances are Z1 and Z2.
Then, according to the first law of electrolysis, we get,
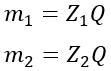
Therefore,
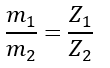
But, the second law of electrolysis states that
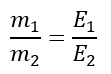
Comparing the above two equations, we get,
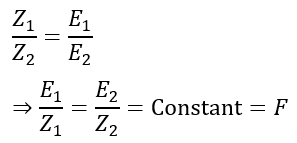
Hence, the ratio of E and Z remains the same for all substances. This constant is known as the Faraday Constant and is denoted by F.
Fundamental Equation of Electrolysis
Let us know how to use the two laws of electrolysis to deduce the fundamental electrolysis equation.
Let us consider an electric charge of Q coulombs passes through the electrolyte, and a mass m is deposited on the cathode. Then, we have,
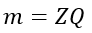
Since,
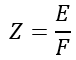
Therefore,
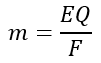
This expression is called the fundamental equation of electrolysis. This equation combines Faraday’s two laws.
Conclusion
In conclusion, this article explains the two fundamental laws of electrolysis, which Michael Faraday gave in 1834. These two laws of electrolysis give the mathematical relationship between the mass of a substance and the electrochemical equivalent and chemical equivalent weight of an element during the electrolysis process. The first law of electrolysis gives the relationship between the mass deposited of the element and the charge passed through the electrolyte. In contrast, the second law of electrolysis gives the relationship between the mass deposited of the element and the chemical equivalent weight of that element.