In this article, we will discuss the important differences between charge and mass. But before that let us first know a bit about charge and mass individually.
Though, charge and mass are the two important properties of matter. The fundamental difference between charge and mass is that charge is related to an electrical property of matter while mass is related to a physical property of matter. Let us discuss more charge and mass.
What is Charge?
Charge, also called electric charge, is the property of matter by virtue of which matter shows electrical behavior. It is the property of subatomic particles namely electrons and protons of the atom. A charge is usually denoted by the symbol Q or q. Its SI unit is Coulomb, represented by C. Based on polarity, the charge is classified into two types, i.e. positive charge and negative charge. The positive charge is carried by protons, whereas the negative charge is carried by electrons.
A charge is quantized, i.e. it always occurs as an integral multiple of fundamental charge (1.6×10-19 C). Also, the charge is conserved, which means the charge neither be created nor destroyed. Another important property of charge is that like charges (+ve and +ve or –ve and –ve) repel each other, whereas unlike charges (+ve and –ve) attract each other.
What is Mass?
Mass is another fundamental property of matter. Mass is the measure of the amount of matter contained by a body. Therefore, mass shows the matter density of the body. The mass of a body is denoted by the symbol m and is measured in kg which is the SI unit of mass. Mass is independent of the place where the body is present. Mass determines the weight of the body at a specific place. Another important property of mass is that it is constant. The mass of an object or body can never be negative. It is also a real number. In the physical analysis of properties of objects, mass plays a vital role.
After discussing the basics of charge and mass, now we are able to discuss the differences between them.
Difference between Charge and Mass
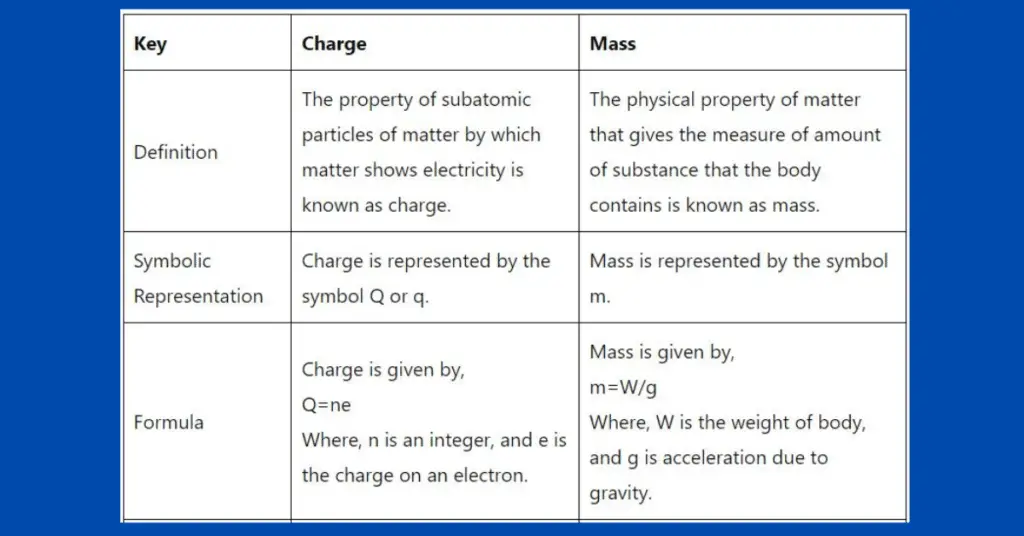
The key differences between charge and mass are given in the table below.
| Key | Charge | Mass |
| Definition | The property of subatomic particles of matter by which matter shows electricity is known as a charge. | The physical property of matter that gives the measure of the amount of substance that the body contains is known as mass. |
| Symbolic Representation | The charge is represented by the symbol Q or q. | Mass is represented by the symbol m. |
| Formula | A charge is given by, Q=ne Where n is an integer, and e is the charge on an electron. | Mass is given by, m=W/g Where, W is the weight of the body, and g is the acceleration due to gravity. |
| SI Unit | The SI unit of charge is Coulomb, denoted by C. | The SI unit of mass is the kilogram, denoted by kg. |
| Type | There are two types of charge namely, positive charge and negative charge. | There is no such classification of mass. Mass is always positive. |
| Nature of Force | The charge can experience a force of attraction as well as repulsion. | Mass always results in a force of attraction. |
| Conservation law | The law of conservation of charge exists. | The law of conservation of mass does not exist. |
| Relativity with speed | The charge is independent of speed. | Mass depends on speed according to the theory of relativity. |
| Dependency | The charge can never exist without mass. It is not necessary that a body must exhibit charge. | Mass can exist without charge. Also, each body present in the universe has a certain mass. |
| Quantization | The charge is quantized as it occurs in integral multiples of electronic charge. | Mass is not quantized, hence mass can be a fractional number as well. |
| Significance | The charge is significant in the analysis of the electrical properties of matter. | Mass is significant in the analysis of the physical properties of matter. |
Conclusion
Hence, this is all about the differences between charge and mass. In conclusion, we can state that charge and mass both are fundamental properties of matter. The study of the above comparison shows that charge and mass have a significant difference that is, the charge can be positive as well as negative, but the mass is always positive.