This article describes the important differences between intrinsic semiconductors and extrinsic semiconductors with their basic description.
Semiconductor devices play a crucial role in designing and fabricating numerous electronic devices due to their peculiar electronic capabilities. A semiconductor is a kind of conducting material whose conducting abilities fall between that of a conductor and an insulator. Semiconductors are temperature and impurities dependant materials.
Based on the conducting capacity, doped impurities, and various other factors, semiconductors are divided into the following two categories.
- Intrinsic Semiconductor
- Extrinsic Semiconductor
What is an Intrinsic Semiconductor?
An intrinsic conductor is a kind of semiconductor material that exists in its highly pure form. In other words, an intrinsic semiconductor is not doped or mixed with any impurity or metallic element. Intrinsic semiconductors are also referred to as i-type semiconductors or undoped semiconductors as well.
At room temperature, intrinsic semiconductors exhibit a negative temperature coefficient of resistance (TCR), which is a measure of the material’s conducting capability. Another peculiar thing to know about an intrinsic semiconductor is that the number of electrons and holes in it are always equal.
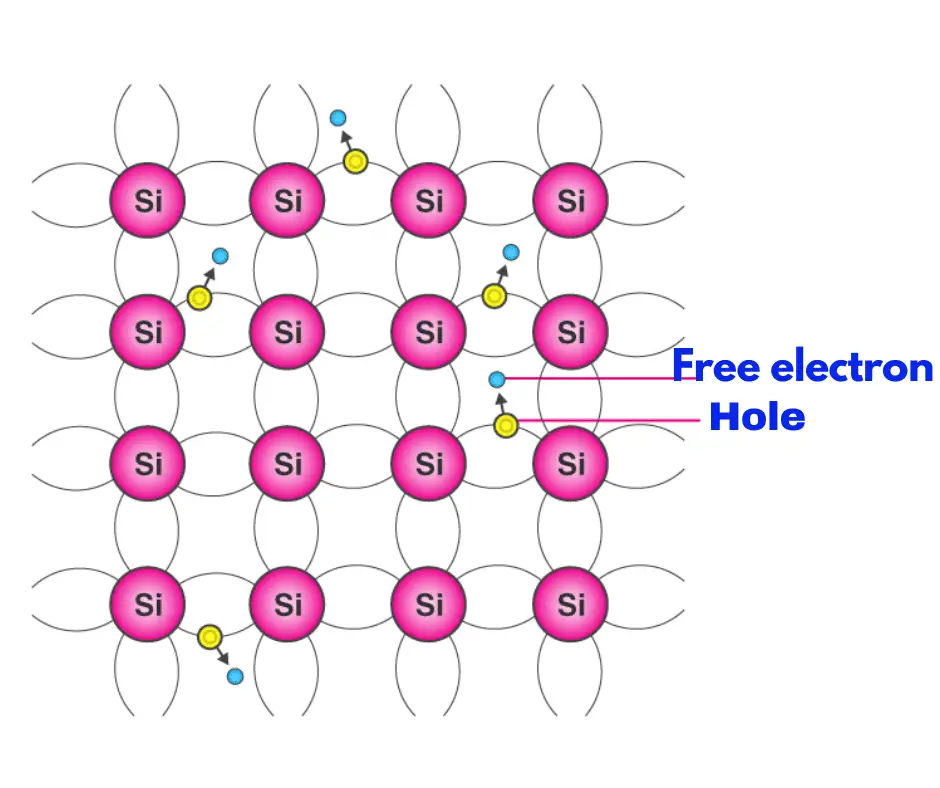
At normal room temperature, an intrinsic semiconductor does not conduct any electricity. But when the intrinsic semiconductor material is exposed to rising temperatures, it shows some signs of conductivity, and with a further increase in temperature, its relative conductivity increases. The elements of Group IV of the periodic table, are all intrinsic conductors in their pure form. But silicon (Si) and Germanium (Ge), out of them all are the most widely used ones.
What is an Extrinsic Semiconductor?
An extrinsic semiconductor is described as a semiconductor material that is doped with external impurity atoms or elements in appropriate amounts, in order to increase its conducting capabilities. The number of holes and electrons is not equal in extrinsic semiconductors. A pure intrinsic semiconductor material can be doped or mixed with elements of group-15 (i.e., elements having 5 valence electrons) or group-13 (i.e., elements having 3 valence electrons).
Based on the type of element being doped, extrinsic semiconductors are subdivided into the following two types.
- N-type semiconductor
- P-type semiconductor
N-type semiconductors are a kind of extrinsic semiconductor material in which a pure form of intrinsic semiconductor such as Silicon (having 4 valence electrons) is doped with pentavalent elements such as Arsenic (As) having five valence electrons. The four valence electrons of tetravalent Silicon form a strong covalent bond with that of the four out of the five valence electrons of Arsenic, resulting in the release of one free electron. The N-type semiconductor has more electrons in comparison to holes.
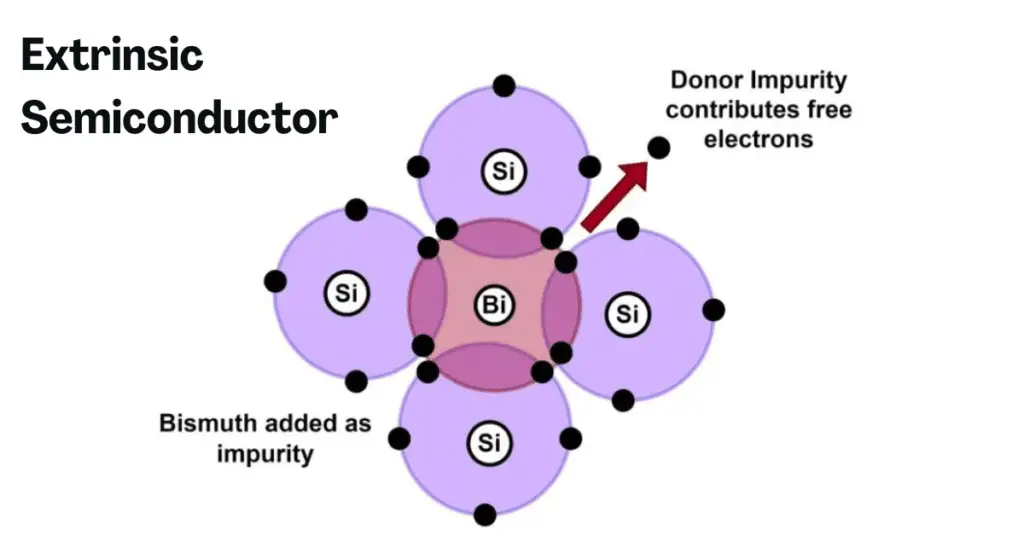
These free electrons result in the semiconductor being rich in negatively charged freely moving electrons, that are responsible for the increased conductivity of the semiconductor material. Furthermore, an increase in the temperatures would result in an increase in the conductivity of the n-type semiconductor material. The P-type semiconductor has more holes compared to electrons.
P-type semiconductors are also a kind of extrinsic semiconductor material in which a pure form of intrinsic semiconductor such as Germanium (having 4 valence electrons) is doped with trivalent elements such as Aluminum (Al) having three valence electrons.
The four valence electrons of tetravalent Germanium form a strong covalent bond with that of three out of the three valence electrons of Aluminum, resulting in the release formation of a hole. These free holes result in the semiconductor being rich in positively charged freely moving holes, which are responsible for the increased conductivity of the semiconductor material.
Furthermore, an increase in the temperatures would result in an increase in the conductivity of the p-type semiconductor material.
Difference Between Intrinsic and Extrinsic Semiconductor
The following table highlights the most notable differences between Intrinsic and Extrinsic types of semiconductor materials.
| Parameter | Intrinsic Semiconductor | Extrinsic Semiconductor |
| Description | Intrinsic semiconductors are a highly pure category of semiconductor materials. | Extrinsic semiconductors are formed by doping the intrinsic semiconductor elements with impurities in regulated amounts. |
| Types of Semiconductors | Intrinsic semiconductors are not further categorized as they are in their purest form already. | Depending on the kind of impurities that the intrinsic semiconductor is doped with, it is divided into the following two categories- P-type and N-type semiconductors. |
| Impurity Doping or mixing | Intrinsic semiconductors are in their form, which means they are not doped or mixed with any kind of impurity element atoms. | Extrinsic semiconductors are doped with impurity trivalent or pentavalent impurity elements. |
| Electric Conductivity | The intrinsic semiconductors have low conducting capability. | Extrinsic semiconductors have relatively high conducting capability. |
| Density of holes / electrons | The number of holes and electrons is the same in both the valence and conduction bands. | The number of holes and electrons differs depending on the properties of the doping material. |
| Factors affecting conductivity | Temperature is the only factor on which the conductivity of the intrinsic semiconductor depends. | Temperature and doping atoms are the two factors on which the conductivity of the extrinsic semiconductor depends. |
| Room temperature conductivity | No conductivity is observed in the case of intrinsic semiconductors at normal room temperature. | Electric conductivity is observed in the case of extrinsic semiconductors at normal room temperature. |
| Conduction temperature | The intrinsic semiconductors start conducting electricity at relatively lower temperatures. | The extrinsic semiconductors start conducting electricity at relatively higher temperatures. |
| Examples | Pure atomic elements such as silicon (Si) and Germanium (Ge) are the most popularly used intrinsic semiconductor materials. | Extrinsic semiconductors are formed by the mixing of either accepting or donating impurity atoms such as Aluminum, Arsenic, Boron, etc. |
Conclusion
In conclusion, this article talks about the main differences between the two broad categories of semiconductors namely – Intrinsic and Extrinsic semiconductors. The most significant difference between them is that Intrinsic semiconductors are pure elements, while extrinsic semiconductors are formed by the mixing or doping of intrinsic semiconductors with impurity atoms.