The Seebeck effect shows the build-up capacity of an electric potential across a temperature gradient in the material. The Seebeck coefficient of the material is a measure of the predominance of the Seebeck effect for a particular type of material.
Let us take a look at the basics of the thermoelectric effect, before discussing the Seebeck effect. Through a thermocouple, the temperature difference across two different metals can be directly converted into electric potential and vice versa.
Thermoelectric effect
A thermoelectric device generates an electric potential when there is a difference in temperature between the junctions on two sides.
In contrast, on the application of electrical potential, heat transfers from one side to the other, causing a temperature difference. An applied temperature gradient causes charge carriers to diffuse from the hot side to the cold side of the material.
The thermoelectric effect can generate electric potential (emf), measure temperature, or change the temperature of an object. Since the direction of applied voltage affects both heating and cooling, thermoelectric devices can be used as temperature controllers.
The term “ Thermoelectric effect” is encircled by three separately identified effects, they are;
- Seebeck effect,
- Peltier effect
- Thomson effect
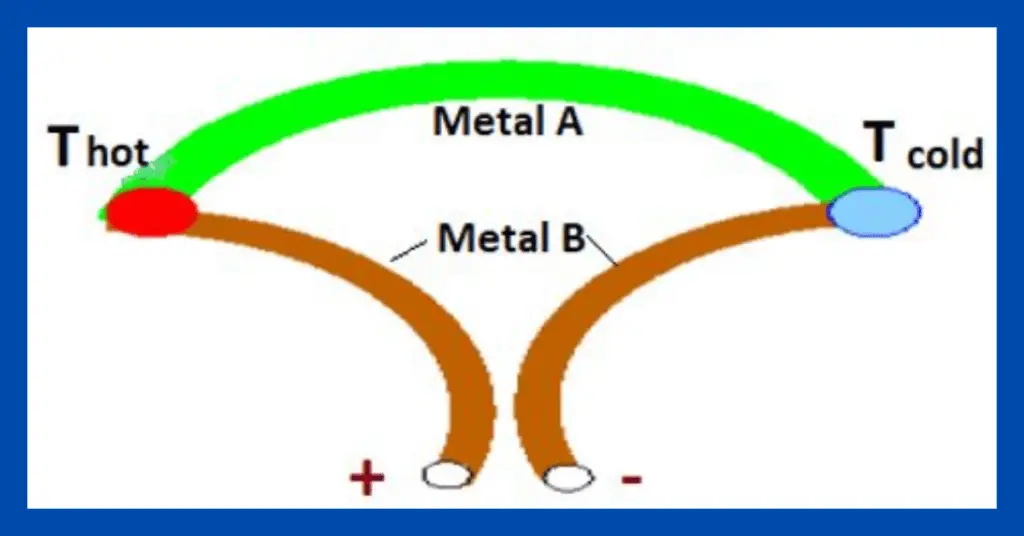
The Seebeck effect describes a thermoelectric phenomenon in which temperature differences between two dissimilar metals or semiconductors in a circuit convert into an electrical voltage.
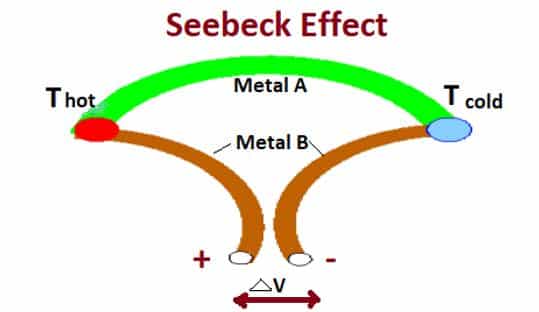
- The Seebeck effect, discovered in 1821, is one of three reversible phenomena that describe similar processes related to temperature, thermoelectricity, and conductivity.
- In the year 1834, the Peltier effect was observed for the first time.
- The Thomson effect was first explained in 1851.
History
The Seebeck effect is named after Thomas Johann Seebeck (1770-831), a German physicist. Seebeck was discovered in 1821. It states that the circuit made of two junctions with two dissimilar metals generates an electromotive force.
When Seebeck moved a compass near the circuit he built and noticed a little drift in the needle. He found that the magnitude of the deviation increased proportionally as the temperature difference increased.
Additionally, his experiments also showed that the temperature distribution along the metallic conductors did not affect the compass. However, changing the types of metals he used changed the magnitude of the needle’s deflection.
During Seebeck’s experiments, the metals reacted to temperatures, creating a current loop in the circuit and a magnetic field. Seebeck was unaware of an electrical current at the time, and wrongly assumed it was the thermo-magnetic effect.
What is the Seebeck effect?
On application of heat to one of the junctions formed by two conductors or semiconductors, causes electrons to dynamize. When one side of the connection experiences this heat, electrons begin to move to the colder side of the two conductors. If the two conductors are in the form of a circuit, a direct current will flow through that circuit.
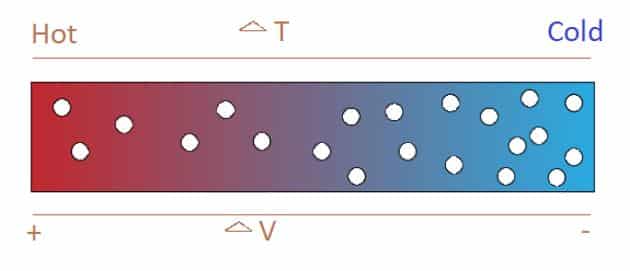
The voltages resulting from the Seebeck effect are very low. However, the range of voltage produced is generally in the order of microvolts (millionths of a volt) per unit temperature. Depending on the temperature difference, some devices can produce a few millivolts.
Several devices can be connected in parallel to increase the capacity of the power supply.
Seebeck Coefficient
The voltage produced by these junctions is proportional to the temperature difference.
The Seebeck coefficient, represented by S, is a proportionality constant. Mathematically, the Seebeck coefficient is presented by the following formula:

The ratio of voltage developed to the temperature difference is related to the intrinsic property of materials termed as “Seebeck coefficient”. It is also popular by the name thermopower or thermoelectric power.
ΔV is the voltage difference.
ΔT is the temperature difference between the two junctions (hot and cold sides). The International System of Units (SI unit) of the Seebeck coefficient is volts per kelvin (V/K). It is also expressed in microvolts per kelvin (µV /K). Seebeck coefficient for semiconductor material is of order a few hundred µV /K. Whereas for metals it is below a hundred only.
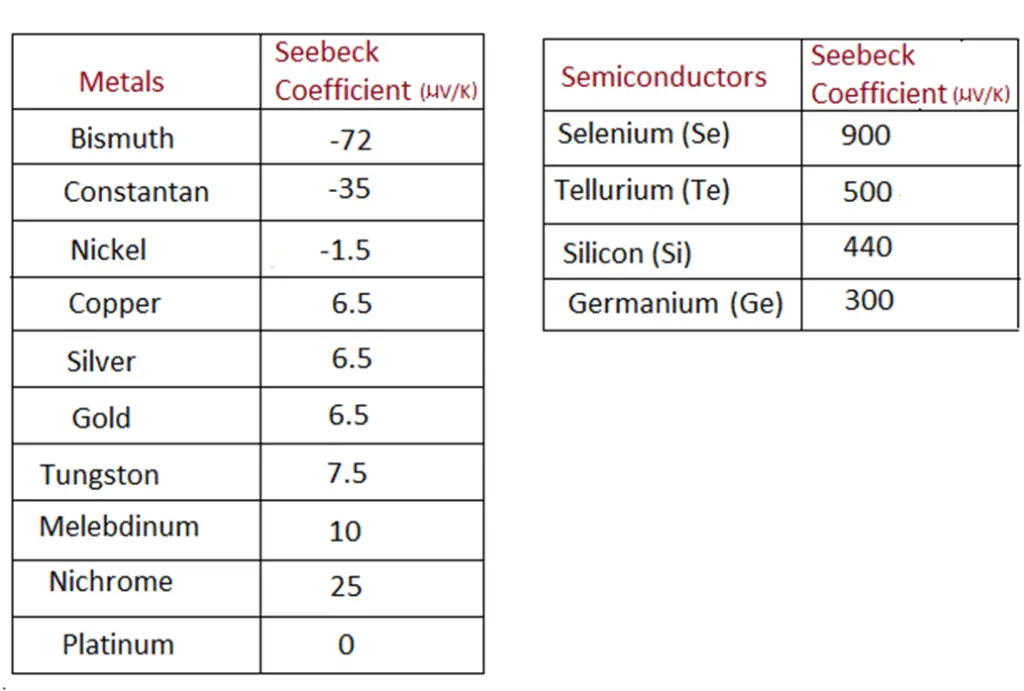
Materials with a high Seebeck coefficient are one among the many factors that contribute to the efficiency of the thermoelectric generators.
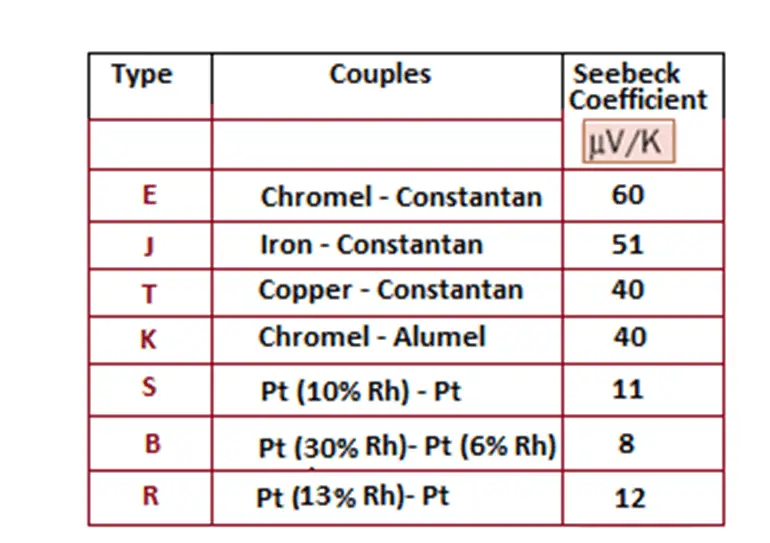
The result of the calculation of the Seebeck coefficient is directly dependent on another factor. Indeed, if the semiconductor material is n-type, the carriers are electrons. In this case, the generated potential difference will be positive, and as for the Seebeck coefficient, it will be negative. If, on the other hand, the semiconductor material is p-type, the potential difference will be negative and therefore the Seebeck coefficient will be positive.
Conductive metals make up a relatively large portion of materials. These metals have different thermoelectric sensitivities, hence different Seebeck coefficients.
Applications of the Seebeck effect
- In general, the Seebeck effect is used in devices called thermocouples that are made by joining two materials, usually metals.
- In thermopiles, several thermocouples are connected in series to increase the output voltage.
- Seebeck effect is the reason for thermoelectric power generation. They have no moving parts and almost maintenance-free operation.
- A temperature difference across any non-superconducting material will create a voltage that can be used to provide energy.
- The pairs of two metals can be used to measure high temperatures.
- In the case of pairs with semiconductors, these are very sensitive to radiation, so radiation thermometers are built with them.
- Also, pairs with semiconductors are efficient detectors of high-frequency electromagnetic waves as well as infrared rays.
Read Next: